Abstract
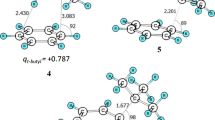
The results of a kinetic study of model reactions between substituted phenols and bromobenzene in the presence of potassium carbonate and the copper(I) chloride–8-hydroxyquinoline catalytic complex in N,N-dimethylformamide are presented. The reactions were found to obey a first-order rate law. Both orbital and charge interactions should be taken into account in a consideration of the effect of the structural characteristics of reactants on the rate of reaction. The kinetics of substitution of the phenoxide anion for an aromatically bound halogen atom in activated and nonactivated systems with the formation of practically valuable aryl ethers were comparatively studied. The results presented provide an opportunity to distinguish the common features and regularities of activated and nonactivated aromatic substitution in the test systems.
Similar content being viewed by others
REFERENCES
USSR Inventor's Certificate no. 1740365, Byull. Izo-bret., 1992, no. 22.
Mil'to, V.I., Orlov, V.Yu., Mironov, G.S., and Kopeikin, V. V., Kinet. Katal., 2001, vol. 42, no.4, p. 518.
Mil'to, V.I., Orlov, V.Yu., Mironov, G.S., and Kopeikin, V. V., Kinet. Katal., 2001, vol. 42, no.4, p. 523.
Pal'm, V.A., Osnovy kolichestvennoi teorii organiches-kikh reaktsii (Bases of Quantitative Theory of Organic Reactions), Leningrad: Khimiya, 1977.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mil'to, V.I., Orlov, V.Y. & Mironov, G.S. Synthesis of Aryl Ethers by Nonactivated Aromatic Nucleophilic Substitution Reactions. Kinetics and Catalysis 44, 747–750 (2003). https://doi.org/10.1023/B:KICA.0000009048.91557.ff
Issue Date:
DOI: https://doi.org/10.1023/B:KICA.0000009048.91557.ff