Abstract
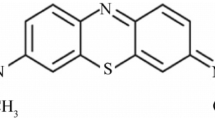
The adsorption conditions of uranium, neptunium, thorium, europium on the adsorbers containing the benzylate salts of the 8-hydroxyquinoline (adsorber B), and of 2-methyl- 8-hydroxyquinoline (adsorber R), were prepared, supported onto charcoal and compared with an adsorber, containing the benzylic acid, (G), stabilized with d-glucoseamine, and adsorbed onto charcoal as well. Thorium, protoactinium, and europium are adsorbed nearly completely from aqueous solutions at a large pH range, even in low acid medium, whereas uranium and neptunium are completely adsorbed only from basic solutions of pH 9. The actinides are preferentially adsorbed with respect to other ions, which are present in natural waters, such as calcium or magnesium. This feature make the analysis of most actinides in natural waters easily to be performed without changing the pH of the original system. The uranium (neptunium) analysis indeed may be accomplished after adjusting the original pH to a fairly basic value (about to 9). The adsorption experiments from real samples showed that the analysis of the actinides dispersed in natural water systems may be successfully performed after previous concentration on the adsorbers prepare.
Similar content being viewed by others
References
M. T. Valentini Ganzerli, V. Crespi Caramella, L. Maggi, J. Radioanal. Nucl. Chem., 260 (2004) 579.
M. T. Valentini Ganzerli, L. Maggi, V. Crespi Caramella, Appl. Radiation Isotopes, 51 (1999) 21.
J. Dervin, J. Faucherre, Bull. Soc. Chim. France, 11 (1973) 2026.
K. J. Raymond, G. E. Freeman, M. J. Kappel, Inorg. Chim. Acta, 94 (1984) 193.
B. Allard, U. Olofsson, B. Torstenfelt, Inorg. Chim. Acta, 94 (1984) 205.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Valentini Ganzerli, M.T., Crespi Caramella, V. & Maggi, L. The role of salts derived from benzilic acid and quinolines in the adsorption of some actinides. Journal of Radioanalytical and Nuclear Chemistry 261, 3–8 (2004). https://doi.org/10.1023/B:JRNC.0000030929.58818.00
Issue Date:
DOI: https://doi.org/10.1023/B:JRNC.0000030929.58818.00