Abstract
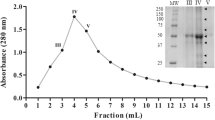
A cysteine endopeptidase, named funastrain c II, was isolated and characterized from the latex of Funastrum clausum (Asclepiadaceae). The molecular mass (mass spectrometry) of the protease was 23.636 kDa. The analysis of funastrain c II by SDS-PAGE revealed a single polypeptide chain. The enzyme showed a remarkable stability of its caseinolytic activity after incubation at temperatures as high as 70°C. Inhibition and activation assays indicated the cysteinic nature of the funastrain c II catalytic site. The optimum pH of funastrain c II enzymatic activity varied according to the substrate used (9.0–10.0 for casein and 6.2–6.8 for PFLNA). Kinetic parameters were determined for N-α-CBZ-Ala p-nitrophenyl ester (K m = 0.0243 mM, k cat = 1.5 s−1) and l-pyroglutamyl-l-phenylalanyl-l-leucine-p-nitroanilide (PFLNA; K M = 0.1011 mM, k cat = 0.9 s−1). The N-terminal sequence of funastrain c II showed considerable similarity to other proteases isolated from latex of different Asclepiadaceae species as well as to other cysteine proteinases belonging to the papain family.
Similar content being viewed by others
References
Abraham, K. I., and Joshi, P. N. (1979a). Biochim. Biophys. Acta 568: 111–119.
Abraham, K. I., and Joshi, P. N. (1979b). Biochim. Biophys. Acta 568: 120–126.
Altschul, S. F., Madden, T. L., Schäffer, A. A., Zhang, J., Zhang, Z., Miller, W., et al. (1997). Nucleic Acids Res. 25: 3389–3402. http://www.ncbi.nlm.nih.gov/blast.
Arribére, M. C., Cortadi, A. A., Gattuso, M. A., Bettiol, M. P., Priolo, N. S., and Caffini, N. O. (1998). Phytochem. Anal. 9: 267–273.
Baker, E. N., and Drenth, J. (1987). In: Jurnak, F., and McPherson, A. R. (eds.), Biological Macromolecules and Assemblies, John Wiley & Sons, New York, pp. 313–368.
Barberis, S., Quiroga, E., Arribére, M. C., and Priolo, N. S. (2000). J. Mol. Catal. B Enzym. 17: 39–47.
Barragán, B. E., Cruz, M. T., del Castillo, L. M., and Castañeda-Agulló, M. (1985). Rev. Latinoamer. Quím. 16: 117–119.
Barret, A. J. (1998). In: Barret, A. J., Rawlings, N. D., and Wocasner, J. F. (eds.), Handbook of Proteolytic Enzymes, Academic Press, London, pp. 546–555.
Barret, A. (2001). In: Beynon, R., and Bond, J. S. (eds.), Proteolytic Enzymes, Second Edition, Oxford University Press, Oxford, pp. 12–17.
Barret, A. J., Kembhavi, A., Brown, M., Kirschke, H., Knight, C. G., Tamai, M., et al. (1982). Biochem. J. 201: 189–198.
Bradford, M. M. (1976). Anal. Biochem. 72: 248–254.
Brockbank, W. J., and Lynn, K. R. (1979). Biochim. Biophys. Acta. 578: 113–122.
Carpenter, D. C., and Lovelace, F. E. (1943). J. Am. Chem. Soc. 65: 2364–2365.
Clapés, P., Torres, J. L., and Aldercreutz, P. (1995). Bioorg. Med. Chem. 3: 245–255.
Conliffe, P. R., Ogilvie, S., Simmen, R. C., Michel, F. J., Saunders, P., and Shiverick, K. T. (1995). Mol. Reprod. Dev. 40: 146–156.
Dubey, K. V., and Jagannadham, M. V. (2003). Phytochemistry 62: 1057–1071.
Dubois, T., Kleinschmidt, T., Schenk, A. G., Looze, Y., and Braunitzer, G. (1988). Biol. Chem. Hoppe-Seyler 369: 741–754.
Englund, P. T., King, T. P., Craig, L. C., and Walti, A. (1968). Biochemistry 7: 163–175.
Filippova, I. Yu., Lysogorskaya, E. N., Oksenoit, E. S., Rudenskaya, G. N., and Stepanov, V. M. (1984). Anal. Biochem. 143: 293–297.
Glaizer, A. N., and Smith, E. L. (1971). In: Boyer, P. D. (ed.), The Enzymes, Academic Press, New York, Vol. III, pp. 501.
Good, N. E., and Izawa, S. (1972). Meth. Enzymol. 24: 53–68.
Gravina de Moraes, M., Termignoni, C., and Salas, C. (1994). Plant Science 102: 11–18.
Greenberg, D. M., and Winnick, T. (1940). J. Biol. Chem. 135: 775–780.
Hansson, A., Veliz, G., Naquira, C. Amren, M. Arroyo, M., and Arevalo, G. (1986). J. Ethnopharmacology 17: 105–138.
Jones, I. K., and Glazer, A. N. (1970). J. Biol. Chem. 245: 2765–2772.
Joseph, L. J., Chang, L. C., Stamenkovich, D., and Sukhatme, V. P. (1988). J. Clin. Invest. 81: 1621–1629.
Kelly, G. (1996). Altern. Med. Rev. 1: 243–257.
Koehler, S. M., and Ho, T. H. (1990). Plant Cell. 2: 769–783.
Kortt, A. A., Hamilton, S., Weeb, E. C., and Zerner, B. (1974a). Biochemistry 13: 2023–2028.
Kortt, A. A., Hinds, J. A., and Zerner, B. (1974b). Biochemistry 13: 2029–2037.
Kramer, D. E., and Whitaker, J. R. (1964). J. Biol. 239: 2178–2183.
Krišstofiková, Z., and Klaschka, J. (1999). Physiol. Res. 48( Suppl. 1): S87.
Kundu, S., Sundd, M., and Medicherla, V. J. (2000). J. Agric. Food Chem. 48: 171–179.
Lynn, K. R., and Yaguchi, M. (1979). Biochim. Biophys. Acta 581: 363–4.
Lynn, K. R., Brockbank, W. J., and Clevette, N. A. (1980). Biochim. Biophys. Acta 612: 119–125.
Mitchel, R. E., Chaiken, I. M., and Smith, E. L. (1970). J. Biol. Chem. 245: 3485–3492.
Obregón, W. D., Arribére, M. C., Morcelle del Valle, S., Liggieri, C., Caffini, N. O., and Priolo, N. S. (2001). J. Protein Chem. 20: 17–25.
Okamura, N., Tamba, M., Uchiyama, Y., Sugita, Y., Dacheux, F., Syntin, P., et al. (1995). Biochim. Biophys. Acta 1245: 221–226.
Pal, G., and Sinha, N. K. (1980). Biochem. Biophys. 202: 321–329.
Perelló, M., Arribére, M. C., Caffini, N. O., and Priolo, N. S. (2000). Acta Farm. Bonaerense 19: 257–262.
Portnoy, D. A., Erickson, A. H., Kochan, J., Ravetch, J. V., and Unkeless, J. C. (1986). J. Biol. Chem. 261: 14697–14703.
Priolo, N. S., López, L. M. I., Arribére, M. C., Natalucci, C. L., and Caffini, N. O. (1991). Acta Alimentaria 20: 189–196.
Priolo, N., Morcelle del Valle, S., Arribére, M. C., López, L. M. I., and Caffini, N. (2000). J. Protein Chem. 19: 39–49.
Robinson, G. W. (1975). Biochemistry 14: 3695–700.
Salvesen, G. S., and Nagase, H. (2001). In: Beynon, R. and Bond, J. S. (eds.), Proteolytic Enzymes, Second Edition, Oxford University Press, Oxford, pp. 116–119.
Schaffer, A. A., Wolf, Y. I., Ponting, C. P., Koonin, E. V., Aravind, L., and Altschult, S. F. (1999). Bioinformatics 15: 1000–1011.
Sengupta, A., Bhattacharya, D., Pal, G., and Sinha, N. K. (1984). Biochem. Biophys. 232: 17–25.
Sgarbieri, V. C., Gupte, S. M., Kramer, D. E., and Whitaker, J. R. (1964). Biol. Chem. 239: 2170–2177.
Shägger, H., and von Jagow, G. (1987). Anal. Biochem. 166: 368–379.
Silverstein, R. M. (1974). Anal. Biochem. 62: 478–484.
Sugiura, M., and Sasaki, M. (1974). Biochem. Biophys. Acta 350: 38–47.
Tablero, M., Arreguín, R., Arreguín, B., Soriano, M., Sánchez, R. I., Rodríguez Romero, A., et al. (1991). Plant Sci. 74: 7–15.
Tatusova, T. A., and Madden, T. L. (1999). FEMS Microbiol. Lett. 174: 247–250; http://embnet.cifn.unam.mx/blast/wblast2.html.
Trejo, S. A., López, L. M. I, Cimino, C. V., Caffini, N. O., and Natalucci, C. L. (2001). J. Protein Chem. 20: 445–453.
Turk, B., Turk, V., and Turk, D. (1997). Biol. Chem. 378: 141–150.
Uhlig, H. (1998). In: Industrial Enzymes and Their Applications. John Wiley & Sons, New York, pp. 146–147.
Vairo Cavalli, S. E., Cortadi, A., Arribére, M. C., Conforti, P., Caffini, N. O., and Priolo, N. S. (2001). Biol. Chem. Hoppe-Seyler 382: 879–883.
Vairo Cavalli, S., Arribére, M. C., Cortadi, A., Caffini, N. O., and Priolo, N. S. (2003). J. Protein Chem. 22: 15–22.
Watanabe, H., Abe, K., Emori, Y., Hosoyama, H., and Arai, S. (1991). J. Biol. Chem. 266: 16897–16902.
Westergaard, J. L., Hackbarth, C., Treuhaft, M. W., and Roberts, R. C. (1980). J. Immunol. Methods 34: 167–175.
Williams, D. C., and Whitaker, J. R. (1969). Plant Physiol. 44: 1574–83.
Winnick, T., Davis, A. R., and Greenberg, D. M. (1940). J. Gen. Physiol. 23: 275–288.
Rights and permissions
About this article
Cite this article
Morcelle, S.R., Trejo, S.A., Canals, F. et al. Funastrain c II: A Cysteine Endopeptidase Purified from the Latex of Funastrum clausum . J Protein Chem 23, 205–215 (2004). https://doi.org/10.1023/B:JOPC.0000026416.90134.7b
Issue Date:
DOI: https://doi.org/10.1023/B:JOPC.0000026416.90134.7b