Abstract
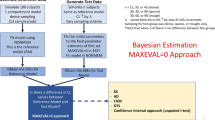
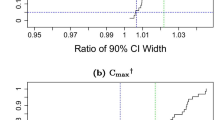
Drug development at different stages may require assessment of similarity of pharmacokinetics (PK). The common approach for such assessment when the difference is drug formulation is bioequivalence (BE), which employs a hypothesis test based on the evaluation of a 90% confidence interval for the ratio of average pharmacokinetic (PK) parameters. The role of formulation effect in BE assessment is replaced by subject population in PK similarity assessment. The traditional approach for BE requires that the PK parameters, primarily AUC and Cmax, be obtained from every individual. Unfortunately in many clinical circumstances, some or even all of the individuals may be sparsely sampled, making the individual evaluation difficult. In such cases, using models, particularly population models, becomes appealing. However, conducting an appropriate statistical test based on population modeling in a form consistent, at least in principle, with traditional 90% confidence interval approach is not so straightforward as it may appear. This manuscript proposes one such approach that can be applied to sparse sampling situations. The approach aims to maintain, as much as possible, the appropriateness of the hypothesis test. It is applied to data from clinical studies to address a need in drug development for assessment of PK similarity in different populations.
Similar content being viewed by others
REFERENCES
D. J. Schuirmann. A comparison of the two one-sided tests procedure and the power approach for assessing the equivalence of average bioavailability. J. Pharmacokinet. Biop-harm. 15 (6): 657–680 (1987).
N. Kaniwa, N. Aoyagi, H. Ogata, and M. Ishii. Application of the NONMEM method to evaluation of the bioavailability of drug products. J. Pharmaceut. Sci. 79 (12): 1116–1120 (1990).
G. A. Maier, G. F. Lockwood, J. A. Oppermann, G. Wei, P. Bauer, and J. Fedler-Kelly. Characterization of the highly variable bioavailability of tiludronate in normal volunteers using population pharmacokinetic methodologies. Eur. J. Drug Metabol. Pharmacokinet. 24 (3): 249–254 (1999).
H. S. Pentikis, J. D. Henderson, N. L. Tran, and T. M. Ludden. Bioequivalence: individ-ual and population compartmental modeling compared to the noncompartmental approach. Pharmaceut. Res. 13 (7): 1116–1121 (1996).
D. M. Reith, W. D. Hooper, J. Parke, and B. Charles. Population pharmacokinetic mod-eling of steady state carbamazepine clearance in children, adolescents, and adults. J. Phar-macokinet. Biop. 28 (1): 79–92 (2001).
S. Senn. Statistical Issues in Drug Development. John Wiley & Son Ltd, 1997.
J. J. Faraway. On the cost of data analysis. J. Comput. Graph. Stat. 1: 213–229 (1992).
C. C. Peck and J. Wechsler. Report of a workshop on con rmatory evidence to support a single clinical trial as a basis for new drug approval. Drug Inf. J. 36: 517–534 (2002).
J. A. Johnson. Inffluence of race or ethnicity on pharmacokinetics of drugs. J. Pharma-ceut. Sci. 86: 1328–1333 (1997).
B. M. Sadler, C. Gillotin, Y. Lou, and D. S. Stein. In vivo effect of alpha-1-acid glyco-protein on pharmacokinetics of amprenavir, a human immunode ciency virus protease inhibitor. Antimicrob. Agents Chemother. 45: 852–856 (2001).
B. Efron and R. J. Tibshirani. An Introduction to the Bootstrap. Chapman & Hall, 1993.
S. L. Beal and L. B. Sheiner. NONMEM User 's Guide. NONMEM project group, UCSF, 1992.
F. Harrell. Regression Modeling Strategies: With Applications to Linear Models, Logistic Regression, and Survival Analysis. Springer Verlag, 2001.
W. R. Gilks, S. Richardson, and D. J. Spiegelhalter. Markov Chain Monte Carlo in Prac-tice. Chapman & Hall, London, 1996.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hu, C., Moore, K.H.P., Kim, Y.H. et al. Statistical Issues in a Modeling Approach to Assessing Bioequivalence or PK Similarity with Presence of Sparsely Sampled Subjects. J Pharmacokinet Pharmacodyn 31, 321–339 (2004). https://doi.org/10.1023/B:JOPA.0000042739.44458.e0
Issue Date:
DOI: https://doi.org/10.1023/B:JOPA.0000042739.44458.e0