Abstract
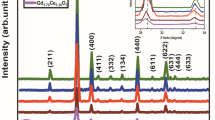
Gadolinium phosphate, obtained through crystallisation from phosphoric acid solution containing Ca-ions, has been characterised by X-ray diffraction, IR-spectra and scanning electron microscopy methods. The presence of Ca-ions in solution resulted in Ca for Gd substitution coupled with simultaneous HPO4 2− incorporation in crystalline hexagonal GdPO4·H2O. The HPO4 2− compensated for the difference in the electric charge between divalent calcium and trivalent gadolinium in the crystallised solid. The amount of Ca in the solid obtained did not exceed 2.5 wt%. Calcium incorporation in gadolinium phosphate caused significant enlargement along the c-axis and less significant contraction along the a-axis of the GdPO4·H2O unit cell.
Similar content being viewed by others
References
S. L. Ng, Y. L. Lam, Y. Zhou, B. S. Ooi, C. H. Kam, K. S. Wong, U. Rambabu and S. Buddhudu, J. Mater. Sci. 37 (2000) 495.
M. Haase, K. Riwotzki, H. Meyssamy and A. Kornowski, J. All. Comp. 303/304 (2000) 191.
J. C. Bourcet and F. K. Fong, J. Chem. Phys. 60 (1974) 34.
N. A. Vasilenko and M. L. Chepelevetski, Z. Neorg. Khim. (Russ) 2 (1957) 2486.
V. I. Sveshnikova and V. L. Ginsburg, ibid. 7 (1962) 1169.
I. V. Tananaev and V. P. Vasilieva, ibid. 8 (1963) 1070.
V. P. Vasilieva, Z. Neorg. Khim. (Russ) Idem., ibid. 9 (1964) 213.
A. I. Ulyanov and T. I. Kazakova, Izvestia Akad. Nauk, USSR, Khimya (Russ) 3 (1963) 393.
Idem., ibid. 7 (1963) 1157.
A. N. Ylyanov, T. I. Kazakova and E. J. Rumiantzeva, ibid. 11 (1962) 1910.
P. A. Bondar, A. I. Domanski, H. Mezentseva, M. G. Degen and N. E. Kalinina, Zh. Neorg. Khim. 21(8) (1976) 2045.
M. Kizilyalli and A. J. E. Welch, J. Appl. Cryst. 9 (1976) 413.
Y. Hikichi, K. Hukuo and J. Shiokawa, Bull. Chem. Soc. Japan 51 (1978) 3645.
J. G. Pepin and E. R. Vance, J. Inorg. Nucl. Chem. 43 (1981) 2807.
R. Kijkowska, J. Mater. Sci. 38 (2003) 229.
R. Kijkowska, E. Cholewka and B. Duszak, ibid. 38 (2003) 223.
R. C. L. Money, Acta Cryst. 3 (1950) 337.
F. Weigel, V. Sherrer and H. Henschel, Radiochim. Acta 4 (1965) 18.
A. Hezel and S. D. Ross, J. Inorg. Nucl. Chem. 29 (1967) 2085.
K. K. Palkina, Neorgan. Mater. 18(9) (1982) 1413.
H. Ito, Y. Fujishiro, T. Sato and A. Okuwaki, British Ceram. Trans. 94 (1995) 146.
K. P. Petrov, I. W. Tananaev, V. G. Pervikh and S. M. Petushkova, Zh. Neorg. Khim. 12 (1967) 2645.
S.-L. Tie, Y-Y. Li and Y.-S. Yang, J. Phys. Chem. Solids 58 (1997) 957.
I. Horvath, I. A. Bondar and L. P. Mezentseva, Zh. Neorg. Khim. (Russ) 31 (1986) 2250.
I. Horvath, I. A. Bondar and L. P. Mezentseva, J. Therm. Anal. 33 (1988) 755.
J. F. W. Bowles and D. J. Morgan, Mineral. Mag. 48 (1984) 146.
R. G. Jonasson, Thermochim. Acta 108 (1986) 65.
Y. Hikichi, T. Sasaki, K. Murayama and T. Nomura, J. Amer. Ceram. Soc. 72 (1989) 1073.
R. Kijkowska, Therm. Acta. 404 (2003) 81.
R. D. Shannon, Acta Cryst. A32 (1976) 751.
O. Terra, N. Clavier, N. Dacheux and R. Podor, New J. Chem. 6 (2003) 957.
Y. Ni and J. M. Hughes, Amer. Mineral. 80 (1995) 21.
R. Z. Legeros, “Calcium Phosphates in Oral Biology and Medicine” (Krager, New York, 1991) p. 95.
R. Kijkowska, S. Lin and R. Z. Legeros, “Bioceramics,” Vol. 14 (http—Trans Tech Publications, Switzerland, 2002) p. 31.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kijkowska, R. Effect of Ca on gadolinium phosphate crystallisation from phosphoric acid solution. Journal of Materials Science 39, 2017–2022 (2004). https://doi.org/10.1023/B:JMSC.0000017763.17037.cf
Issue Date:
DOI: https://doi.org/10.1023/B:JMSC.0000017763.17037.cf