Abstract
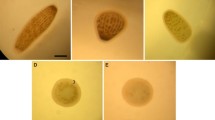
The effects of the chemical inducers, gamma-aminobutyric acid (GABA) and potassium chloride (KCl), on the larval settlement and metamorphosis of the donkey-ear abalone, Haliotis asinina, was investigated. H. asinina larvae (5–6 h post-hatch) were exposed to a range of GABA (0.125–2.00 μM) and KCl (1.00–12.00 mM) concentrations for 72 h. Results of the dose response experiments showed that settlement and metamorphosis vary according to the dose levels of the inducer compounds. Under controlled laboratory conditions, 0.45–0.50 μM and 6.0 mM seemed to be the optima for GABA and KCl, respectively, as these concentrations elicited the greatest number of postlarvae that metamorphosed, settled or survived. However, GABA generally promoted better attachment and metamorphic response as well as survival than KCl in H. asinina postlarvae.
Similar content being viewed by others
References
Akashige, S., T. Seki, H. Kan-no & T. Nomura, 1981. Effects of gamma aminobutyric acid and certain neurotransmitters on the settlement and metamorphosis of the larvae of Haliotis discus hannai Ino (Gastropoda). Bulletin of Tohoku Fisheries Research Laboratory 43: 37–35.
Barlow, L. A., 1990. Electrophysiological and behavioral responses of larvae of the red abalone (Haliotis rufescens) to settlementinducing substances. Bulletin of Marine Science 46(2): 537–554.
Bryan, P. J. & P.-Y. Qian, 1998. Induction of larval settlement and metamorphosis in the abalone, Haliotis diversicolor (Reeve). Journal of Experimental Marine Biology and Ecology 223(1): 39–51.
Fermin, A. C. & R. S.J. Gapasin, 2000. Postlarvae density and photoperiod effects on the settlement and metamorphosis of the donkey-ear abalone, Haliotis asinina (Linne, 1758). Proceedings of the 10th Tropical Marine Mollusc Programme (TMMP) Congress and Workshop, 19-31 October 1999, Hanoi, Vietnam. Phuket Marine Biological Center Special Publication 21(1): 231–234.
Hahn, K. O., 1989. Handbook of Culture of Abalone and Other Marine Gastropods. CRC Press, Inc., Boca Raton, FL, 348 p.
Jarayabhand, P. & N. Paphavasit, 1996. A review of the culture of tropical abalone with special reference to Thailand. Aquaculture 140: 159–168.
Kaspar, H. F. & D. O. Mountfort, 1995. Microbial production and degradation of ?-aminobutyric acid (GABA) in the abalone larval settlement habitat. FEMS Microbiology and Ecology 17: 205–212.
Kawamura, T., 1996. The role of benthic diatoms in the early life stages of the Japanese abalone (Haliotis discus hannai). Proceedings of an International Workshop on Survival Strategies in Early Life Stages of Marine Resources. A.A. Balkema, Rotterdam: 355–367.
Morse, D. E., 1984. Biochemical and genetic engineering for improved production of abalone and other valuable molluscs. Aquaculture 39: 263–282.
Morse, D. E., N. Hooker, H. Duncan & L. Jensen, 1979a. ?-aminobutyric acid, a neurotransmitter, induces planktonic abalone larvae to settle and begin metamorphosis. Science 204: 407–410.
Morse, D. E., N. Hooker, L. Jensen & H. Duncan, 1979b. Induction of larval settling and metamorphosis by ?-aminobutyric acid and its congeners from crustose red algae. II. Applications to cultivation, seed-production and bioassays; principal causes of mortality and interference. Proceedings of theWorld Mariculture Society 10: 81–91.
Morse, A. N. C. & D. E. Morse, 1984. Recruitment and metamorphosis of Haliotis larvae induced by molecules uniquely available at the surfaces of crustose coralline algae. Journal of Experimental Marine Biology and Ecology 75: 191–215.
Moss, G.A., 1999. Factors affecting settlement and early postsettlement survival of the New Zealand abalone Haliotis australis. New Zealand Journal of Marine and Freshwater Research 33: 271–278.
Roberts, R. D., 2001. A review of settlement cues for larval abalone (Haliotis spp.). Journal of Shellfish Research 20(2): 571–586.
Roberts, R. D. & C. M. Nicholson, 1997. Variable response from abalone larvae (Haliotis iris, H. virginea) to a range of settlement cues. Molluscan Research 18: 131–141.
Sawatpeera, S., E. Suchart Upatham, M. Kruatrachue, Y. P. Chitramvong, P. Sonchaeng, T. Pumthong & J. Nugranad, 2001. Larval development in Haliotis asinina Linnaeus. Journal of Shellfish Research 20(2): 593–601.
SAS Institute, 1988. SAS/STAT TM User’s Guide, Release 6.03 Edition. SAS Institute, Cary, North Carolina, USA, 1028 p.
Searcy-Bernal, R. & C. Anguiano-Beltran, 1998. Optimizing the concentration of gamma-aminobutyric acid (GABA) for inducing larval metamorphosis in the red abalone, Haliotis rufescens (Mollusca: Gastropoda). Journal of the World Aquaculture Society 29(4): 463–470.
Searcy-Bernal, R., A. E. Salas-Garza, R. A. Flores-Aguilar & P. R. Hinojosa-Rivera, 1992. Simultaneous comparison of methods for settlement and metamorphosis induction in the red abalone, Haliotis rufescens. Aquaculture 105: 241–250.
Slattery, M., 1992. Larval settlement and juvenile survival in the red abalone (Haliotis rufescens): an examination of inductive cues and substrates selection. Aquaculture 102: 143–153.
Singhagraiwan, T. & M. Doi, 1993. Seed production and culture of a tropical abalone, Haliotis asinina Linne. Eastern Marine Fisheries Development Center (EMDEC), Thailand and Japan International Cooperation Agency (JICA), 32 pp.
Singhagraiwan, T. & M. Sasaki, 1991. Breeding and early development of the donkey’s ear abalone Haliotis asinina Linne. Thai Marine Fisheries Research Bulletin, 2: 95–100.
Yang, Y. & B.-L. Wu, 1995. Induction of larval settlement and metamorphosis of Haliotis discus hannai Ino (Gastropa, Mollusca). Chinese Journal of Oceanology and Limnology 13(1): 71–77.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gapasin, R.S., Polohan, B.B. Induction of larval settlement and metamorphosis in the donkey-ear abalone, Haliotis asinina Linnaeus, by chemical cues. Hydrobiologia 519, 9–17 (2004). https://doi.org/10.1023/B:HYDR.0000026480.00928.5a
Issue Date:
DOI: https://doi.org/10.1023/B:HYDR.0000026480.00928.5a