Abstract
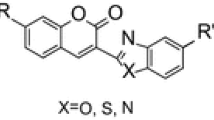
A stepwise sequence for the interaction of 3-ethoxycarbonyl(acyl)coumarins with cyanoacetylhydrazine, its N-acetyl and N-isopropylidene derivatives, leading to the formation of 3-cyanocoumarins, is proposed and demonstrated. It was established that the 3-cyanocoumarins formed are also able to participate in further conversions by a type of Michael reaction.
Similar content being viewed by others
REFERENCES
V. D. Nageswara Sastry andT. R. Seshadri, Proc. Indian Acad. Sci., 16A, 29 (1942); Chem. Abstr., 37, 880 (1943).
A. Sammour,M. Abdalla, andM. Elkady, Acta Chim. Hung., 82, 369 (1974).
M. P. Nemeryuk,V. D. Dimitrova,A. L. Sedov, andV. F. Traven, Khim. Geterotsikl. Soedin., 1417 (1997).
V. F. Traven,V. D. Dimitrova,A. L. Sedov,R. V. Rozhkov,M. P. Nemeryuk,M. R. Salem, andE. A. Carberry, Heterocycl. Commun., 4, 33 (1998).
P. B. Terentiev andA. P. Stankyavichus, Mass Spectrometric Analysis of Biologically Active Nitrogen Bases [in Russian], Mokslas, Vilnius (1987), p. 149.
C. N. O'Callaghan, J. Chem. Soc., C, 207 (1971).
P. Czerney andH. Hartmann, J. Prakt. Chem., 323, 691 (1981).
Abdel-Kerim M. N. Gohar,F. F. Abdel-Latif, andM. S. El-Ktatny, Indian J. Chem., 25B, 404 (1986).
H. Junek andF. Frosch, Z. Naturforsch., 26B, 1124 (1971).
N. A. Ismail,F. A. Khalifa, andA. A. Magd El Din, Heterocycles, 32, 1101 (1991).
R. Clinging,F. M. Dean, andL. E. Houghten, J. Chem. Soc., C, 897 (1970).
E. C. Horning andM. G. Horning, J. Am. Chem. Soc., 69, 968 (1947).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nemeryuk, M.P., Dimitrova, V.D., Anisimova, O.S. et al. Conversion of Coumarins Accompanied by Opening and Recyclization of the Lactone Ring. 1. Study of the Reaction of 3-Ethoxycarbonyl(3-acyl)coumarins with Cyanoacetylhydrazine and Its Derivatives. Chemistry of Heterocyclic Compounds 39, 1454–1465 (2003). https://doi.org/10.1023/B:COHC.0000014410.73636.dd
Issue Date:
DOI: https://doi.org/10.1023/B:COHC.0000014410.73636.dd