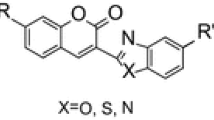
Abstract
The regioselectivity of a one-pot hydrogenation/alkylation reaction of coumarins 1a and 1b with series of alkyl halides has been elucidated as a complementary work to reaction of acylation with the same coumarins. Reaction conditions have provided formation of only C-alkylation products with good to high yield. The results from the experiments are of significant importance to predict reactivity of 3-substituted coumarins in different reaction conditions.

Similar content being viewed by others
References
Bojilova A, Ivanov C (1986) Synthesis of the nitrile and some esters of 3,3-dimethyl-2-oxochroman-4-acetic acid. Synthesis 5:415–416. https://doi.org/10.1055/s-1986-31658
Bojilova A, Nikolova R, Ivanov C, Rodios NA, Terzis A, Raptopoulou CP (1996) A comparative study of the interaction of salicylaldehydes with phosphonoacetates under Knoevenagel reaction conditions. Synthesis of 1,2-benzoxaphosphorines and their dimers. Tetrahedron 52:12597. https://doi.org/10.1016/0040-4020(96)00748-X
Gomez-Bengoa E, Landa A, Lizarraga A, Mielgo A, Oiarbide M, Palomo C (2011) Catalytic asymmetric α-alkylation of aldehydes via a SN2′-type addition-elimination pathway Chem. Science 2:353–357. https://doi.org/10.1039/c0sc00402b
Ho TS (1975) The hard and soft acids and bases (HSAB) principle and organic chemistry. Chem Rev 75:1. https://doi.org/10.1021/cr60293a001
Höfle G, Steglich W, Vorbrüggen H (1978) 4-Dialkylaminopyridines as highly active acylation catalysts. Angew Chem Int Ed Engl 17:569–583. https://doi.org/10.1002/anie.197805691
Ilieva ED, Petkova NI, Nikolova RD (2012) A new and efficient method for the synthesis of 3,4-disubstituted pyrrolidine-2,5-diones. Molecules 17:4936. https://doi.org/10.3390/molecules17054936
Janecki T, Kedzia J, Wasek T (2009) Michael addition to activated vinylphosphonates. Synthesis 8:1227. https://doi.org/10.1055/s-0028-1088031
Kadin SB (1966) Reduction of conjugated double bonds with sodium borohydride. J Org Chem 31(1):620–622. https://doi.org/10.1021/jo01340a523
Kirkiacharian BS, Danan A (1986) Reductions via boranes: a new, convenient method for the preparation of 3-substituted esters and thioesters of 3,4-dihydrocoumarin. Synthsis. https://doi.org/10.1055/s-1986-31643
Koleva AI, Petkova-Yankova NI, Nikolova RD (2016) Ultrasound-assisted conjugate addition of organometallic reagents to 3-diethylphosphonocoumarin. Synlett 27:2676. https://doi.org/10.1055/s-0036-1589167
Koleva AI, Petkova-Yankova NI, Nikolova RD (2018) Ultrasound assisted metal-mediated method for the formation of tetrahydro-3,3′-disubstituted biscoumarins. Molecules 23:2810. https://doi.org/10.3390/molecules23112810
Koleva AI, Petkova-Yankova NI, Nikolova RD (2019) Synthesis and chemical properties of 3-phosphono-coumarins and 1,2-benzoxaphosphorins as precursors for bioactive compounds molecules. Molecules 24(11):2030–2068. https://doi.org/10.3390/molecules24112030
Minammi T, Motoyoshiya J (1992) Vinylphosphonates in organic synthesis. Synthesis. https://doi.org/10.1055/s-1992-26103
Mitra AK, Misra SK, Patra A (1980) New synthesis of 3-Alkyl coumarins. Synth Commun 10:915–919. https://doi.org/10.1080/00397918008061851
Pearson RG, Songstad J (1967) Application of the principle of hard and soft acids and bases to organic chemistry. JACS. https://doi.org/10.1021/ja00984a014
Petkova NI, Nikolova RD, Bojilova AG, Rodios NA, Raptopoulou CP (2006) Hydrogenation/regioselective C-acylation reaction of diethyl coumarin-3-phosphonate with NaBH4/acid anhydrides. A new one pot tandem reaction. Synth Commun 36:509. https://doi.org/10.1080/00397910500385167
Petkova NI, Nikolova RD, Bojilova AG, Rodios NA, Kopf J (2009) Synthesis of heterocyclic methylene bisphosphonates by 1,3-dipolar cycloaddition of ethyl diazoacetate to 1,2-benzoxaphosphorin-3-phosphonates. Tetrahedron 65:1639. https://doi.org/10.1016/j.tet.2008.12.048
Petkova NI, Nikolova RD, Kostov KL, Mineva T, Vayssilov GN (2014) Theoretical and experimental local reactivity parameters of 3-substituted coumarin derivatives. J Phys Chem A 118:11062. https://doi.org/10.1021/jp508432c
Petkova-Yankova NI, Nikolova RD (2018) Decarboxylation of β-ketochromanes to β-ketophosphonates; collection of articles from the National Scientific Conference “15 Years Pharmacy in Medical University Plovdiv” ISBN 978-619-237-014-5 (online)
Sakakura A, Kawajiri K, Ohkubo T, Kosugi Y, Ishihara K (2007) Widely useful DMAP-catalyzed esterification under auxiliary base- and solvent-free conditions. J Am Chem Soc 129:14775–14779. https://doi.org/10.1021/ja075824w
Savignac P, Bogdan I (2003) Modern phosphonate chemistry. CRC Press LLC, London
Scriven EFV (1983) 4-Dialkylaminopyridines: super acylation and alkylation catalysts. Chem Soc Rev 12:129–161. https://doi.org/10.1039/CS9831200129
Vieth S, Costisella B, Schneider M (1997) Tandem michael addition alkylation of vinylphosphonates. Tetrahedron 53:9623. https://doi.org/10.1016/S0040-4020(97)00648-0
Wamhoff H, Schorn G, Korte F (1967) Zur synthese und Umlagerung bi- und tricyclischer α-Acyl-δ-lactone. Chem Ber 100(4):1296–1304. https://doi.org/10.1002/cber.19671000431
Acknowledgements
This work was supported by National Science Fund project "Dimeric coumarin structures – new synthetic approaches and quantum-chemical investigations" - KP-06-N-39/15. The authors would like to express their gratitude to Prof. Nestor A. Rodios from Laboratory of Organic Chemistry, Department of Chemistry, Aristotle University of Thessaloniki, for the NMR and MS spectra.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Petkova-Yankova, N.I., Nikolova, R.D. Substituted coumarins as ambident nucleophiles in one-pot hydrogenation/alkylation reaction. Chem. Pap. 74, 2627–2634 (2020). https://doi.org/10.1007/s11696-020-01098-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01098-1