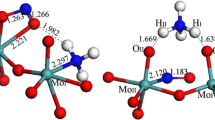
Abstract
The influence of conditions of the preliminary thermal treatment of ZrO2, ammonia and methanol adsorption, and MoO3 supporting on O2 – formation during the adsorption of an NO + O2 mixture was studied. The interaction of O2 – with different molecules was studied. Adsorbed ammonia and methanol, as well as supported Mo6+ ions, were shown to inhibit this reaction. The involvement of the Zr4+ and O2– Lewis sites in the reaction was concluded. The interaction of ammonia and methanol with the O2 – radical anions changed the g tensor parameters and decreased the thermal stability of O2 – in the case of methanol. O2 – radical anions were formed on the reduced (0.1–2.0)% MoO3/ZrO2 samples during the interaction of O2 with the Mo5+ ions in the octahedral configuration. As in the case of O2 – formation during NO + O2 adsorption on ZrO2, the radical anions were localized in the coordination spheres of the coordinately unsaturated Zr4+ ions. A change in the MoO3 content of the samples from 0.1 to 0.5% led to an increase in the amount of O2 –, whereas a change from 0.5 to 2.0% led to a decrease in the O2 – amount due to the screening of the Zr4+ ions by oxo complexes and polymolybdates.
Similar content being viewed by others
REFERENCES
Occhiuzzi, M., Giambelli, P., Sammino, D., Ghiotti, G., and Prinetto, F., Stud. Surf. Sci. Catal, 1996, vol. 101A, p. 691.
Delahay, G., Ensuque, E., Cog, B., and Figueras, F., J. Catal., 1998, vol. 175,no. 1, p. 7.
Indovina, V., Campa, M.C., and Tuti, D., 4th Eur. Congr. on Catalysis, Italy, 1999, p. 268.
Konin, G.A., Il'ichev, A.N., Matyshak, V.A., and Korchak, V.N., Mendeleev Commun., 2000, no. 5, p. 197.
Il'ichev, A.N., Konin, G.A., and Matyshak, V.A., Kulizade, A.M., Korchak, V.N., and Yan, Yu.B., Kinet. Katal., 2002, vol. 43,no. 2, p. 235.
Chuah, G.K., Catal. Today, 1999, vol. 49, p. 131.
Chen, S.X.K., Bell, A.T., and Iglesia, E., J. Phys. Chem. B, 2000, vol. 104, p. 1059.
Tret'yakov, I.I., Shub, B.R., and Sklyarov, A.V., Zh. Fiz. Khim., 1970, vol. 44, p. 2112.
Handbuch der Praparativen Anorganischen Chemie in Drei Banden, Herausgegeben Von G. Brauer, Ed., Stuttgart: Ferdinand Enke, 1981.
Che, M. and Tench, A.J., Adv. Catal., 1983, vol. 32, p. 1.
Garrone, E., Giamello, E., Ferraris, M., and Spoto, G., J. Chem. Soc., Faraday Trans., 1992, vol. 88,no. 3, p. 333.
Fenin, V.M., Shvets, V.A., and Kazanskii, V.B., Kinet. Katal., 1971, vol. 12,no. 5, p. 1255.
Narishige, N. and Niwa, M., Catal. Lett., 2001, vol. 71,nos. 1–2, p. 63.
Bhaskar, T., Reddy, K.R., Kumar, C.R., Murthy, M., and Chary, K., Appl. Catal., A, 2001, vol. 211, p. 189.
Abdo, S., LoJacono, M., Crarkson, R.B., and Holl, W.K., J. Catal., 1975, vol. 36, p. 330.
Kucherov, A.V. and Slinkin, A.A., Usp. Khim., 1992, vol. 61,no. 9, p. 1687.
Aramendia, M.A., Borau, V., Jimenez, C., Marinas, J.M., Marinas, A., Porras, A., and Urbano, F.J., J. Catal., 1999, vol. 183, p. 240.
Krylov, O.V., Pariisky, G.B., and Spiridonov, K.N., J. Catal., 1971, vol. 23, p. 301.
Maity, S.K., Rana, M.S., Srinivas, B.N., Bej, S.K., Dhar, G.V., and Rao, T.S.R., J. Mol. Catal., A: Chem., 2000, vol. 153, p. 121.
Mikheikin, I.D., Zhidomirov, G.M., and Kazanskii, V.B., Usp. Khim., 1972, vol. 41,no. 5, p. 909.
Vorotyntsev, V.M., Shvets, V.A., and Kazanskii, V.B., Kinet. Katal., 1971, vol. 12,no. 5, p. 1249.
Abdo, S., Clarkson, R.B., and Hall, W.K., J. Phys. Chem., 1976, vol. 80,no. 21, p. 2431.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Il'ichev, A.N., Shibanova, M.D. & Korchak, V.N. The ESR Study of O2 – Radical Anion Formation during Adsorption of an NO + O2 Mixture and O2 on ZrO2 and (0.1–2.0)% MoO3/ZrO2 . Kinetics and Catalysis 45, 114–121 (2004). https://doi.org/10.1023/B:KICA.0000016111.58075.5f
Issue Date:
DOI: https://doi.org/10.1023/B:KICA.0000016111.58075.5f