Abstract
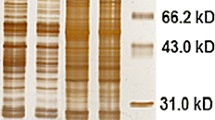
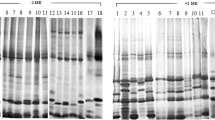
Proteins from pollen and leaves of parent forms and of a hybrid and an amphiploid Aegilops geniculata × S. cereale were subjected to a study by two-dimensional gel electrophoresis (2-DE). The majority of peptides found in the parents were revealed in the amphiploid pollen and the hybrid and amphiploid leaves, however, numerous parental peptides were absent both in hybrid and amphiploid. There were detected characteristic peptides in leaves of the hybrid as well as in the leaves and pollen of the amphiploid, which were absent in the material from both parents. 52 peptides were common for pollen and leaves of the amphiploid what corresponds to 14.2% and 25.7% of peptides in pollen and leaves, respectively. Three characteristic peptides were common for leaves and pollen of the amphiploid (MW 48.0 kDa in pH 5.9, MW 49.0 kDa in pH 6.9 and MW 22.0 kDa in pH 8.8).
Similar content being viewed by others
References
Apolinarska, B., 1996. Transfer of chromosomes of the A and B genomes of wheat to tetraploid rye. J Appl Genet 37: 345–356.
Aragoncillo, C., M.A. Rodriguez-Loperena, G. Salcedo, P. Carbonero & F. Garcia-Olmedo, 1978. Influence of homeologus chromosomes on gene-dosage effects in allohexaploid wheat (Triticum aestivum). Proc Natl Acad Sci USA 75: 1446–1450.
Chen, W.-T., 1992. Membrane proteases: roles in tissue remodeling and tumor invasion. Curr Opin Cell Biol 4: 802–809.
Colas des Francs, C. & H. Thiellement, 1985. Chromosomal localisation of structural genes and regulators in wheat by 2D electrophoresis of ditelosomic lines. Theor Appl Genet 71: 31–38.
Dickinson, H.G., C.J. Eleman & J. Doughty, 2000. Pollen coatingchimeric genetics and new funcions. Sex Plant Reprod 12: 302–309.
Doughty, J., S. Dixon, S.J. Hiscock A.C. Willis, I.A.P. Parkin & H.G. Dickinson, 1998. PCP-A1, a defensin like Brassica pollen coat protein that binds the S locus glycoprotein, is the product of gametophytic gene expression. Plant Cell 10: 1333–1347.
Doughty, J., F. Hedderson, A. McCubbin & H. Dickinson, 1993. Interaction between a coating-borne peptide of Brassica pollen grain and stigmatic S (self-incompatibility)-locus specific glycoproteins. Proc Natl Acad Sci USA 90: 467–471.
Doughty, J., H.Y. Wong & H.G. Dickinson, 2000. Cysteine-rich pollen coat proteins (PCPs) and their interaction with stigmatic S (incompatibility) and S-related proteins in Brassica: putative roles in SI and pollination. Ann Bot 85(A): 161–169.
Galili, G. & M. Feldman, 1984. Intergenomic suppression of endosperm protein genes in common wheat. Can J Genet Cytol 26: 651–656.
Heukeshoven, J. & R. Dernick, 1985. Simplified method for silver staining of proteins in polyacrylamide gels and the mechanism of silver staining. Electrophoresis 6: 103–112.
Hochstrasser, D.F., M.G. Harrington, A.C. Hochstrasser, M.J. Miller & C.M. Merril, 1988. Methods for increasing the resolution of two-dimensional protein electrophoresis. Anal Biochem 173: 424–435.
Howlett, B.J., R.B. Knox, J.B. Pactons & J. Heslop-Harrison, 1975. Pollen-wall proteins: Physiochemical characterisation and role in self-incompatibility in Cosmos bibinatus. Proc R Soc London B 188: 167–182.
Hurkman, W.J. & Ch.K. Tanaka, 1986. Solublization of plant membrane proteins for analysis by two-dimensional gel electrophoresis. Plant Physiol 81: 802–806.
Jordá, L., A. Coego, V. Conejero & P. Vera, 1999. A genomic containing four differentially regulated subtilisin-like procesing protease genes is in tomato plants. J Biol Biochem 274: 2360–2365.
Kalinowski, A., K. Winiarczyk & B. Wojciechowska, 2001. Pollen proteins after two-dimensional gel electrophoresis and pollen morphology of the amphiploids Aegilops kotschyi and Ae. variabilis with Secale cereale. Sex Plant Reprod 14: 153–161.
Kalinowski, A., B. Wojciechowska & E. Borzyszkowska, 2003. Isozymes in Aegilops kotschyi and Ae. biuncialis × Secale cereale hybrids and Ae. kotschyi × S. cereale amphiploids in relation to their parents. J Appl Genet 44: 35–43.
Kimber, G., P.J. Sallee & M.M. Feiner, 1988. The interspecific and evolutionary relationships of Triticum ovatum. Genome 30: 218–221.
Kimber, G. & Y. Yen, 1988. Analysis of pivotal-differential evolutionary patterns. Proc Natl Acad Sci USA 85: 9106–9108.
Kimber, G. & Y. Yen, 1989. Hybrids involving wheat relatives and autotetraploid Triticum umbellulatum. Genome 32: 1–5.
Knox, R.B. & J. Heslop-Harrison, 1970. Pollen wall proteins: localisation and enzymatic activity. J Cell Sci 6: 1–27.
Lukaszewski, A., B. Apolinarska, J.P. Gustafson & K.D. Krolow, 1984. Chromosome constitution of tetraploid triticale. Z Planzenzücht 93: 222–236.
Luu, D.T., P. Heizmann & Ch. Dumas, 1997. Pollen-stigma adhesion in kale is not depend on the self-(in) compatibility genotype. Plant Physiol 115: 1221–1230.
Marais, G.F. & A.S. Marais, 1994. The derivation of compensating translocation involving homeologus group 3 chromosomes of wheat and rye. Euphytica 79: 75–80.
Mulcahy, D.M., 1974. Correlation between speed of pollen tube growth and seedling height in Zea mays. Nature 249: 491–492.
Nettancourt, D. de, 2001. Incompatibility and Incongruity in Wild and Cultivated Plants. Springer-Verlag, Berlin, Heildelberg, New York, pp. 236–243.
Ottaviano, E. & D.L. Mulcahy, 1989. Genetic of angiosperm pollen. Adv Genet 26: 1–64.
Ozkan, H., A.A. Levy & M. Feldman, 2001. Allopolyploidyinduced rapid genome evolution in the wheat (Aegilops-Triticum) group. Plant Cell 13: 1735–1747.
Shaked, H., K. Kashkush, H. Ozkan, M. Feldman & A.A. Levy, 2001. Sequence elimination and cytosine methylation are rapid and reproductible responses of the genome to wide hybridization and alloplyploidy in wheat. Plant Cell 13: 1749–1759.
Tanskley, S.D., D. Zamir & Ch.M. Rick, 1981. Evidence for extensive overlap of sporophytic and gametophytic gene expression in Lycopersicon esculentum. Science 213: 453–455.
Thiellement, H., N. Bahrman & C. Colas des Francs, 1986. Regulatory effects of homeologus chromosome arms on wheat proteins at two developmental stages. Theor Appl Genet 73: 246–251.
Tornero, P., V. Conejero & P. Vera, 1996. Primary structure and expression of a pathogen-induced protease (PR-P69) in tomato plants: similarity of funcional domains to subtilisin-like endoproteases. Proc Natl Acad Sci USA 93: 6332–6337.
Tornero, P., V. Conejero & P. Vera, 1997. Identification of a new pathogen-induced member of the subtilisin-like processing protease family from plants. J Biol Biochem 272: 14412–14419.
Wojciechowska, B. & H. Pudelska, 1999. Production, morphology and fertility of the amphiploids Aegilops variabilis × Secale cereale and Ae. kotschyi × S. cereale. Cereal Res Commun 27: 79–82.
Wojciechowska, B. & H. Pudelska, 2002. Hybrids and amphiploids of Aegilops ovata L. with Secale cereale L.: production, morphology and fertility. J Appl Genet 43: 415–421.
Zivy, M., P. Devaux, J. Blaisonneau, R. Jean & H. Thielement, 1992. Segregation distortion and linkage studies in microspore-derived double haploid lines Hordeum vulgare L. Theor Appl Genet 83: 919–924.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kalinowski, A., Wojciechowska, B. Pollen and leaf proteins after 2-D electrophoresis of the Aegilops geniculata × Secale cereale hybrids, amphiploids and parental forms. Euphytica 133, 201–207 (2003). https://doi.org/10.1023/A:1025578310370
Issue Date:
DOI: https://doi.org/10.1023/A:1025578310370