Abstract
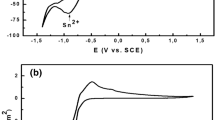
A bath for the electrodeposition of white gold alloys of interest for the electroforming of hollow jewellery is proposed and investigated. The system was an acidic Au(III)–Sn(IV) bath for the electrodeposition of Au–Sn alloys. The electrochemical investigations were based on cyclic voltammetry, linear-sweep voltammetry, galvanostatic electrodeposition experiments and in situ Raman spectroscopy. The electrode kinetics of alloy formation were elucidated by stripping voltammetry. The effects of cathodically adsorbed CN− were studied by in situ Raman spectroscopy. Electrodeposited foils were studied from the crystallographic, compositional and morphological points of view. Codeposition of Au and Sn gives rise to a single phase of approximately equiatomic composition over a current density interval of 10 to 40 mA cm−2. This orthorhombic phase is structurally the same as the ζ′ phase of the equilibrium Au–Sn system, but its stoichiometry and lattice parameters are different. The equilibrium two-phase δ, ζ′ structure can be obtained by heat-treatment.
Similar content being viewed by others
References
B. Bozzini and P.L. Cavallotti, Trans. IMF 78 (2000) 227.
P. Bagnoud, S. Nicoud and P. Ramoni, Gold Technology 18 (1996) 11.
B. Bozzini, AIFM Galvanotecnica e Nuove Finiture 3 (1993) 199.
B. Bozzini, P.L. Cavallotti, G. Giovannelli and S. Natali, Elettrocristallizzazione di leghe binarie Au-Sn a composizione stechiometrica, XX Congresso Nazionale della Societa` Chimica Italiana, Rimini, 4-9 giugno, 2000, EL-OR003.
B. Bozzini, P.L. Cavallotti, G. Giovannelli and S. Natali, ‘Electrodeposition And Structural Characterisation Of Au-Sn Alloys’, Proceedings of the 15th World INTERFINISH Congress, 13-15 Sept. (2000), Garmisch-Partenkirchen, Germany.
V.A. Vicente and S. Bruckenstein, Anal. Chem. 44 (1972) 297.
A. Rodes, J.M. Feliu, A. Aldaz and J. Clavilier, J. Electroanal. Chem. 256 (1988) 455.
A. Rodes, E. Herrero, J.M. Feliu and A. Aldaz, J. Chem. Soc., Faraday Trans. 92 (1996) 3769.
W. Zilske, Electrolytic bath and process for the deposition of gold alloy coatings, US Patent 4 391 679; P. Stevens, Tin-gold electroplating bath and process, US Patent 4 013 523; F. Zantini, Electrodeposition of gold alloys, US Patent 3 764 489.
W. Sun and D.G. Ivey, Mat. Sci. Eng. B65 (1999) 111.
B. Bozzini, G. Giovannelli and P.L. Cavallotti, J . Appl. Electrochem. 29 (1999) 685.
B. Bozzini, G. Giovannelli and P.L. Cavallotti, J. Appl. Electrochem. 30 (2000) 591.
F.R. Brotzen. Intern. Mat. Rev. 39 (1994) 24.
B. Bozzini and A. Fanigliulo, J. Appl. Electrochem, in press.
C. Korzeiewski, S. Pons, P.P. Schmidt and M.W. Severson, J. Chem. Phys. 85 (1986) 4153.
D.K. Lambert, J. Chem. Phys. 89 (1988) 3847.
J.S. Luo, R.G. Tobin, D.K. Lambert, G.B. Fisher and C.L. DiMaggio, J. Chem. Phys. 99 (1993) 1347.
S.A. Wasileski and M.J. Weaver, Faraday Discuss. 121 (2002) 285.
K. Ashley, F. Weinert and D.L. Feldheim, Electrochim. Acta 36 (1991) 1863.
S. Zou and M.J. Weaver, J. Phys. Chem. 100 (1996) 4237.
D.K. Lambert, Electrochim. Acta 41 (1996) 623.
T. Iwasita and F.C. Nart, in H. Gerischer and C.W. Tobias (Eds), ‘Advances in Electrochemical Science and Engineering’ (VCH, Weinheim, 1995), p. 123.
A.B. Anderson, R. Kötz and E. Yeager, Chem. Phys. Lett. 82 (1981) 130.
B. Bozzini, Faraday Discuss. 121 (2002) 231, 345.
C.Y. Chen, E. Burstein and S. Lundquist, Solid State Comm. 32 (1979) 63.
K. Osada, S. Yamaguchi and M. Hirabayashi, Trans. Jpn. Inst. Met. 15 (1974) 256.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bozzini, B., Fanigliulo, A., Giovannelli, G. et al. Electrodeposition of Au–Sn alloys from acid Au(III) baths. Journal of Applied Electrochemistry 33, 747–754 (2003). https://doi.org/10.1023/A:1025079215480
Issue Date:
DOI: https://doi.org/10.1023/A:1025079215480