Abstract
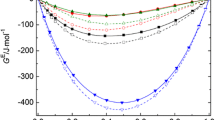
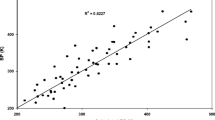
Comparison of the boiling points (Tb) of the members of the organic triglyceride and anhydride homologous series (with unbranched hydrocarbon chains) with the Tb of n-alkanes at equal Van der Waals molecular volumes showed that the boiling points of compounds with functional groups, centers of strong intermolecular interactions, are lower than those of hydrocarbons, compounds containing no functional groups. In the triglyceride and anhydride homologous series, the Tb sharply (by more than 100°C) decreases from homolog to homolog (rather than increasing, as usually observed). To explain these anomalies, computer modeling of conformations of individual and associated molecules was performed. Standard enthalpies of vaporization (ΔH v 0), energies of intermolecular interactions (EIMI), and the ratios EIMI/ΔH v 0 = αIMI (coefficients of intermolecular interactions, which may serve as a criterion of the fraction of such interactions, converted into intramolecular interactions) were calculated for all the compounds. The Tb of triglycerides and anhydrides calculated in terms of the model of pair reversible interactions of like atomic groups with allowance for αIMI nicely fit with experimental values. The resulting data show that part of intermolecular interactions in triglycerides and anhydrides converts into intramolecular, which results in anomalously low Tb and sharp fall of Tb along the triglyceride and anhydride homologous series.
Similar content being viewed by others
REFERENCES
Korolev, G.V., Il'in, A.A., Sizov, E.A., and Mogilevich, M.M., Zh. Obshch. Khim. 1999, vol. 69, no. 10, p. 1630.
Korolev, G.V., Il'in, A.A., Sizov, E.A., and Mogilevich, M.M., Zh. Obshch. Khim. 1999, vol. 69, no. 10, p. 1636.
Molecular Mechanics Burkert, U. and Allinger, N.L., Eds., Washington, DC: ACS, 1982.
Korolev, G.V., Il'in, A.A., Chebunin, R.V., Solov'ev, M.E., and Mogilevich, M.M., Izv. Vyssh. Uchebn. Zaved. Khim. Khim. Tekhnol. 2001, no. 6, p. 6.
Dictionary of Organic Compounds Heilbron, J. and Bunbury, H. M., Eds., London: Eyre and Spottiswoode, 1946, vols. 1-3.
Lange, M., Handbook of Chemistry New York: Interscience, 1959.
Chemist's Hanbook Nikol'skii, B.P., Ed., Leningrad: Khimiya, 1963, vol. 2.
Kitaigorodskii, A.I., Organicheskaya kristallokhimiya (Organic Crystallochemistry), Moscow: Akad. Nauk SSSR, 1955.
Askadskii, A.A. and Matveev, Yu.I., Khimicheskoe stroenie i fizicheskie svoistva polimerov (Chemical Structure and Physical Properties of Polymers), Moscow: Khimiya, 1983.
Korolev, G.V., Il'in, A.A., Sizov, E.A., Solov'ev, M.E., and Mogilevich, M.M., Zh. Obshch. Khim. 2000, vol. 70, no. 7, p. 1088.
Korolev, G.V. and Berezin, M.P., Vysokomol. Soedin., Ser. A 1997, vol. 39, no. 2, p. 242.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Korolev, G.V., Il'in, A.A., Mogilevich, M.M. et al. Peculiar Features of Intermolecular Interactions in Organic Compounds with Anomalously Low Boiling Points. Russian Journal of General Chemistry 73, 334–339 (2003). https://doi.org/10.1023/A:1024937315410
Issue Date:
DOI: https://doi.org/10.1023/A:1024937315410