Abstract
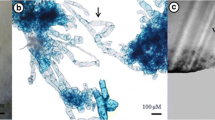
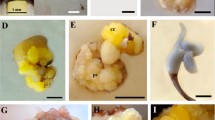
Regeneration in oil palm was achieved through somatic embryogenesis/organogenesis from embryo-derived callus. Callus was induced from mature embryos of the cross 281 (D)×18 (P) on modified MS medium supplemented with 2,4-D (113.12 μM) and 2-iP (14.76 μM). The embryogenic calluses obtained were transferred to Blaydes medium supplemented with 2,4-D (0.045 μM) and one of the following growth regulators: TDZ (4.54 μM), zeatin riboside (2.85 μM), putrescine (1 mM) and spermine (100 μM). Secondary somatic embryogenesis was found to occur in media supplemented with polyamines. The efficiency of formation of somatic embryos, secondary somatic embryos and shoot meristemoids were significantly higher in putrescine containing medium. Histological studies were also undertaken.
Similar content being viewed by others
References
Aberlenc-Bertossi F, Noirot M & Duval Y(1999) BA enhances the germination of oil palm somatic embryos derived from embryogenic suspension cultures. Plant Cell Tiss. Org. Cult. 56: 53–57
Adkins SW, Samosir YM, Ernawati A, Godwin ID & Drew RA (1998) Control of ethylene and use of polyamines can optimise the conditions for somatic embryogenesis in coconut (Cocos nucifera L.) and papaya (Carica papaya L.). Acta Horti. (ISHS) 461: 459–466
Anitha K & Sajini KK (1996) Plantlet regeneration from leaf explants of oil palm. Curr. Sci. 71: 922–926
Blaydes DF (1966) Interaction of kinetin and various inhibitors in the growth of soybean callus. Physiol. Plant. 19: 748–753
Besse I, Verdeil J-L, Duval Y, Sotta B, Maldiney R & Miginia E (1992) Oil palm (Elaeis guineensis Jacq.) clonal fidelity: endog-enous cytokinins and indolacetic acid in embryogenic callus cultures. J. Exp. Bot. 43: 983–989
Chi GL, Lin WS, Lee JEE & Pua EC (1994) Role of polyamines on de novo shoot morphogenesis from cotyledons of Brassica campestris ssp. pekinensis (Lour) Olsson in vitro. Plant Cell Rep. 13: 323–329
Duval Y, Engelmann F & Durand-Gasselin T Somatic embryogenesis in oil palm (Elaeis guineensis Jacq ). In: Bajaj YPS (ed) Somatic Embryogenesis and Synthetic Seeds I - Biotechnology in Agriculture and Forestry Vol. 30 (pp. 335–352). Springer Verlag, Berlin, Heidelberg, New York
Feray A, Hourmant A, Brun A & Penot M (1993) Effects of polyamines on morphogenesis of in vitro potato plants (Solanum tuberosum cv. Bintje). Compte-de-l'Academic-des-Sciences-Serie-III-Science-de-la-vie 316: 1446–1451
Fienberg AA, Choi JH, Lubich WP & Sung ZR (1984) Develop-mental regulation of polyamine metabolism in growth and differentiation of carrot culture. Planta 162: 532–539
Graille J & Pina M (1999) The role of palm oil in the human diet. Plant. Recherche Dev. 6: 91–93
Galston AW & Flores H E (1991) Polyamines and plant mor-phogenesis. In: Slocum RD & Flores HE (eds) Biochemistry and Physiology of Polyamines in Plants., CRC Press, London
Galston AW & Kaur-Sawhney R (1990) Polyamines in plant physiology. Plant. Physiol. 94: 406–410
Huetteman CA & Preece JE (1993) Thidiazuron: a potent cytokinin for woody plant tissue culture. Plant Cell Tiss. Org. Cult. 33: 105–119
Jones LH (1990) Endogenous cytokinins in oil palm (Elaeis guineensis Jacq.) callus, embryoids and regenerant plants mea-sured by radioimmunoassay. Plant Cell Tiss. Org. Cult. 20: 201–209
Jones LH, Hanke DE & Eeuwens CJ (1995) An evaluation of the role of cytokinins in the development of abnormal inflorescences in oil palm (Elaeis guineensis Jacq.) regenerated from tissue culture. J. Plant Growth Regul. 914: 135–142
Kevers C, le Gal N, Monteiro M, Dommes J, Gaspar T & le Gal N (2000) Somatic embryogenesis of Panax ginseng in liquid cultures: a role for polyamines and their metabolic pathways. Plant Growth Regul. 31: 209–214
Martin-Tanguy J & Carre M (1993) Polyamines in grapevine microcuttings cultivated in vitro - effects of amines and inhibitors of polyamine biosynthesis on polyamine levels and microcutting growth and development. Plant Growth Regul. 13: 269–280
Martinez LE, Aguero CB, Lopez ME & Galmarini CR (2000) Improvement of in vitro gynogenesis induction in onion (Allium cepa L.) using polyamines. Plant Sci. 156: 221–226
Muhitch M J, Edwards LA & Fletcher JS (1983) Influence of diamines and polyamines on the senescence of plant suspension culture. Plant Cell Rep. 92: 82–84
Murashige T & Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 915: 473–497
Murthy BNS, Murch SJ & Saxena PK (1998) Thidiazuron: A potent regulator of in vitro plant regeneration. In Vitro Cell. Dev. Biol. Plant 34: 267–275
Parthasarathy U, Parthasarathy VA & Karun A (2001) Oil palm. In: Parthasarathy VA, Bose TK, Deka PC, Das P, Mitra SK & Mohandas S (eds) Biotechnology of Horticultural Crops Vol. 1 (pp. 595–608).
Naya Prokash, Calcutta Raju CR, Sajini KK, Balachandran SM, Saji KV, Geetha Maheshan K, Rajasekharan PE, Geetha L & Ahamed Bavappa KV (1989) Clonal multiplication of oil palm (Elaeis guineensis Jacq.). J. Plant. Crops. 16: 17–20
Sargent HR, Godwin ID & Adkins SW (1998) The effect of exogenous polyamines on somatic embryogenesis and plant regeneration from Sorghum bicolor and Saccharum spp. Acta Horti. (ISHS) 461: 451–458
Snedecor GW & Cochran RW (1975) Statistical Methods. Oxford and IBH Publishing Co, New Delhi
Smith TA (1985) Polyamines. Annu. Rev. Plant Physiol. 36: 117–143
Yadav JS & Rajam MV (1997) Spatial distribution of free and conjugated polyamines in leaves of Solanum melogena L. associated with differential morphogenetic capacity: efficient somatic embryogenesis with putrescine. J. Exp. Bot. 48: 1537–1545
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rajesh, M., Radha, E., Karun, A. et al. Plant regeneration from embryo-derived callus of oil palm – the effect of exogenous polyamines. Plant Cell, Tissue and Organ Culture 75, 41–47 (2003). https://doi.org/10.1023/A:1024679910085
Issue Date:
DOI: https://doi.org/10.1023/A:1024679910085