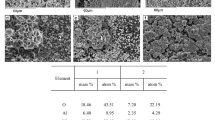
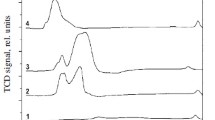
Abstract
Catalytic systems composed of copper-based oxides and alkali alkoxides are tested for low-temperature methanol synthesis in liquid phases, which involves carbonylation of methanol to methyl formate and consecutive hydrogenation of methyl formate to methanol. The effects of reaction variables on the catalytic performance are investigated under the conditions of 373-423K temperature and 1.5-5.0 Mpa pressure. The combined catalytic systems of copper chromite and potassium methoxide exhibit excellent activities for the production of methanol. Higher values of reaction temperature, initial pressure, catalyst loading, and H2/CO ratio of the feed gas lead to higher methanol productivity. In particular, the reaction temperatures and the feed gas compositions strongly influence the catalytic performance. No methanol is formed when employing a feed gas containing 2% CO2. The catalytic systems are deactivated in a short period even in a CO2-free feed gas, due to the consumption of the alkoxide component. Alkali alkoxides are consumed through reactions with trace amounts of CO2 and H2O which are formed as by-products during the course of the runs. The results also suggest that the hydrogenation step of methyl formate over copper chromite is greatly accelerated in the presence of the alkali alkoxide.
Similar content being viewed by others
References
D.L. Trimm and M.S. Wainwright, Catal. Today 6 (1990) 261.
M. Marchionna, in: Oxygenates by Homologation or CO Hydrogenation with Metal Complexes, ed. G. Braca (Kluwer Academic Publishers, Dordrecht, 1994) p. 57.
S. Ohyama, PETROTECH 18 (1995) 27.
S. Ohyama, Stud. Surf. Sci. Catal. 130 (2000) 3753.
R.S. Sapienza, W.A. Slegeir, T.E. O'Hare and D. Mahajan, US Patent 4 614 749 (1986).
R.S. Sapienza, W.A. Slegeir, T.E. O'Hare and D. Mahajan, US Patent 4 619 946 (1986).
R.S. Sapienza, W.A. Slegeir, T.E. O'Hare and D. Mahajan, US Patent 4 623 634 (1986).
H. Nakamura, K. Saeki and M. Tanaka, Jpn. Patent 88/51129 (1988).
S.T. Sie, E. Drent and W.W. Jager, US Patent 4 812 433 (1989).
M. Marchionna, M. Lami, F. Ancillotti and R. Ricci, Ital. Patent 20028/A (1988).
M. Marchionna, L. Basini, A. Aragno, M. Lami and F. Ancillotti, J. Mol. Catal. 75 (1992) 147.
S. Ohyama, Appl. Catal. A 180 (1999) 217.
S. Ohyama, Appl. Catal. A 181 (1999) 87.
O.T. Onsager, Jpn. Patent 87/500867 (1987).
O.T. Onsager, Jpn. Patent 91/12048 (1991).
Z. Liu, J.W. Tierney, Y.T. Shah and I. Wender, Fuel Process. Tech. 18 (1988) 185.
Z. Liu, J.W. Tierney, Y.T. Shah and I. Wender, Fuel Process. Tech. 23 (1989) 149.
V.M. Palekar, H. Jung, J.W. Tierney and I. Wender, Appl. Catal. A 102 (1993) 13.
V.M. Palekar, J.W. Tierney and I. Wender, Appl. Catal. A 103 (1993) 105.
S.P. Tonner, D.L. Trimm, M.S. Wainwright and N.W. Cant, J. Mol. Catal. 18 (1983) 215.
I. Wender, Catal. Rev. Sci. Eng. 26 (1984) 303.
D.J. Darensbourg, R.L. Gray and C. Ovalles, J. Mol. Catal. 29 (1985) 285.
D.J. Darensbourg, R.L. Gray and C. Ovalles, J. Mol. Catal. 41 (1987) 329.
R.J. Gormley, A.M. Giusti, S. Rossini and V.U.S. Rao, 9th Int. Congr. Catal. 2 (1988) 553.
P.Å. Sørum and O.T. Onsager, 8th Int. Congr. Catal. 2 (1984) 233.
K.M. Kim, H.C. Woo, M. Cheong, J.C. Kim, K.H. Lee, J.S. Lee and Y.G. Kim, Appl. Catal. A 83 (1992) 15.
D.M. Monti, M.A. Kohler, M.S. Wainwright, D.L. Trimm and N.W. Cant, Appl. Catal. 22 (1986) 123.
S.P. Tonner, D.L. Trimm and M.S. Wainwright, Ind. Eng. Prod. Res. Dev. 23 (1984) 384.
R.J. Gormley, V.U.S. Rao, Y. Soong and E. Micheli, Appl. Catal. A 87 (1992) 81.
D.M. Monti, M.S. Wainwright, D.L. Trimm and N.W. Cant, Ind. Eng. Chem. Prod. Res. Dev. 24 (1985) 397.
D.M. Monti, N.W. Cant, D.L. Trimm and M.S. Wainwright, J. Catal. 100 (1986) 17.
D.M. Monti, N.W. Cant, D.L. Trimm and M.S. Wainwright, J. Catal. 100 (1986) 28.
J.A. Christiansen, U.S. Patent 1 302 011 (1919).
M. Imaizumi, in: Shokubai Koza Vol. 8, ed. Catalysis Society of Japan (Kodansha Scientific), Tokyo, 1985) p. 3.
S. Ohyama and H. Kishida, Appl. Catal. A 172 (1998) 241.
S. Ohyama and H. Kishida, Stud. Surf. Sci. Catal. 121 (1999) 431.
S. Ohyama and H. Kishida, Appl. Catal. A 184 (1999) 239.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ohyama, S. Low-Temperature Methanol Synthesis in Catalytic Systems Composed of Copper-Based Oxides and Alkali Alkoxides in Liquid Media: Effects of Reaction Variables on Catalytic Performance. Topics in Catalysis 22, 337–343 (2003). https://doi.org/10.1023/A:1023500725571
Issue Date:
DOI: https://doi.org/10.1023/A:1023500725571