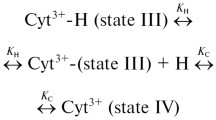Abstract
The molten globule state (MG) of cytochrome c is the major intermediate of protein folding. The formation of MG state of cytochrome c is induced by n-alkyl sulfates such as sodium octyl sulfate (SOS), sodium dodecyl sulfate (SDS), and sodium tetradecyl sulfate (STS). The folding state of cytochrome c was monitored using circular dichroism (CD), isothermal titration calorimetry (ITC) and partial specific volumes. To explore a new approach for characterizing the MG conformation, cyclic voltametric studies of n-alkyl sulfates induced transition at acidic pH of cytochrome c (unfolded state, U) was carried out. Here, we have used a cystein-modified gold electrode, which is effective for direct rapid electron transfer to cytochrome c even in acid solutions, to directly observe electrochemistry in native (N) cytochrome c. Our results show that the extent of electron transfer is increased for U→MG, and also the easiness of electron transferring occurred from MG→N transition. Thus we demonstrate that the MG state of cytochrome c, induced by n-alkyl sulfates as salts with hydrophobic chains (hydrophobic salts), with different compactness reaches to near identical amount of electron transferring as N state.
Similar content being viewed by others
References
Arakawa, T., and Timasheff, S. N. (1982). Biochemistry 21: 6536–6544.
Babul, J., and Stellwagen, E. (1973). Can. J Biochem. 51: 465–471.
Barker, P. D., and Mauk, A. G. (1992). J. Am. Chem. Soc. 114: 3619–3624.
Bathaie, S. Z., Moosavi-Movahedi, A. A., and Saboury, A. A. (1999). Nucleic Acids Res. 27: 1001–1005.
Bixler, J., Bakker, G., and McLendon, G. (1992). J. Am. Chem. Soc. 114: 6938–6939.
Bushnell, G. W., Lovie, G. V., and Brayer, G. D. (1990). J. Mol. Biol. 214: 585–595.
Christensen, H., and Pain, R. H. (1991). Biophys. J. 19: 221–229.
Elove, G. A., Bhuyan, A. K., and Roder, H. (1994). Biochemistry 33: 6925–6935.
Ferri, T., Poscia, F., Ascola, F., and Santucci, R. (1996). Biochim. Biophys. Acta 1298: 102–108.
Fisher, W. R., Taniuchi, H., and Anfinsen, C. B. (1973). J. Biol. Chem. 248: 3188–3195.
Goto, Y., and Fink, L. (1989). Biochemistry 28: 945–952.
Goto, Y., and Nishikori, S. (1991). J. Mol. Biol. 222: 679–686.
Goto, Y., Calciano, L. J., and Fink, A. L. (1990a). Proc. Natl. Acad. Sci. USA 87: 573–577.
Goto, Y., Takahashi, N., and Fink, L. A. (1990b). Biochemistry 29: 3480–3488.
Griko, Y. V., and Privalov, P. L. (1994). J. Mol. Biol. 235: 1318–1325.
Hamada, D., Hoshino, M., Kataoka, M., Fink, A. L., and Goto, Y. (1993). Biochemistry 32: 10351–10358.
Hamada, D., Kidokoro, S., Fukada, H., Takahashi, K., and Goto, Y. (1994). Proc. Natl. Acad. Sci. USA 91: 10325–10329.
Huange, Z. X., Feng, M., Wang, Y. H., Cui, J., and Zho, D. S. (1996). J. Electroanal Chem. 416: 31–40.
Hyakawa, K., Saterre, J. P., and Kwak, J. C. T. (1983). Biophys. Chem. 17: 175–181.
Jeng, M. F., Englander, S. W., Elove, G. A., Wand, A. J., and Roder, H. (1990). Biochemistry 29: 10433–10437.
Jones, M. N., Finn, A., Moosavi-Movahedi, A. A., and Waller, B. J. (1987). Biochim. Biophys. Acta. 91: 3395–3398.
Kamiyama, T., Sadahide, Y., Nogusa, Y., and Gekko, K. (1999). Biochim. Biophys. Acta 1434: 44–57.
Kataoka, M., Hagihara, Y., Mihata, K., and Goto, Y. (1993). J. Mol. Biol. 229: 591–596.
Kataoka, M., Nishii, I., Fujisawa, T., Ueki, T., Tokunaga, F., and Goto, Y. (1995). J. Mol. Biol. 249: 215–228.
Kim, P. S., and Baldwin, R. L. (1990). Annu. Rev. Biochem. 59: 631–660.
Kuroda, Y., Kidokoro, S., and Wada, A. (1992). J. Mol. Biol. 223: 660–679.
Kuwajima, K. (1986). Proteins 6: 87–103.
Lojou, E., and Bianco, P. (2000). J. Electroanal Chem. 485: 71–80.
Lovie, G. V., Pielak, G. J., Smith, M., and Brayer, G. D. (1988). Biochemistry 27: 7870–7880.
Nishii, I., Kataoka, M., and Goto, Y. (1995). J. Mol. Biol. 250: 223–238.
Nishii, I., Kataoka, M., Tokonaga, F., and Goto, Y. (1994). Biochemistry 33: 4903–4909.
Ohgushi, M., and Wada, A. (1983). FEBS Lett. 164: 21–24.
Pande, A., and Meyer, C. A. (1978). Biochem. Biophys. Res. Commun. 85: 7–11.
Pielak, G. J., Concar, D. W., Moore, G. R., and Williams, R. J. P. (1987). Protein Eng. 1: 83–88.
Pineda, T., Selvilla, J. M., Roman, A. J., and Blazquez, M. (1997). Biochim. Biophys. Acta 1343: 227–234.
Potekhin, S., and Pfeil, W. (1989). Biophys. Chem. 34: 55–62.
Ptitsyn, O. B. (1987). J. Protein. Chem. 6: 273–293.
Ptitsyn, O. B. (1992). The molten globule state, in Creighton, T. E. (Ed.), Protein folding, W. H. Freeman and Co., New York, pp. 243–300.
Roder, H., and Elove, G. A. (1994). Early stages of protein folding, In mechanisms of protein folding (Pain, R. H., ed.), Oxford University Press, New York, pp. 26–54.
Reimer, D. L., Zhang, Y. P., Kong, S., Wheeler, J. J., Graham, R. W., and Bally, M. B. (1995). Biochemistry 34: 12877–12883.
Roder, H., Elove, G. A., and Englander, S. W. (1988). Nature 335: 700–704.
Sawyer, D. T., Sobkowiak, A., and Roberts, J. L. (1995). Electrochemistry for Chemists, 2nd ed., John Wiley & Sons, New York.
Sosnick, T. R., Mayne, L., Hiller, R., and Englander, S. W. (1994). Nat. Struct. Biol. 1: 149–156.
Takakuwa, T., Konno, T., and Meguro, H. (1985). Anal. Sci. 1: 215–218.
Udgaonkar, J. B., and Baldwin, R. L. (1998). Nature 335: 694–699.
Zhou, Y., Nagaoka, T., and Zhu, G. (1999). Biophys. Chem. 79: 55–62. —-
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moosavi-Movahedi, A.A., Chamani, J., Ghourchian, H. et al. Electrochemical Evidence for the Molten Globule States of Cytochrome c Induced by N-Alkyl Sulfates at Low Concentrations. J Protein Chem 22, 23–30 (2003). https://doi.org/10.1023/A:1023011609931
Published:
Issue Date:
DOI: https://doi.org/10.1023/A:1023011609931