Abstract
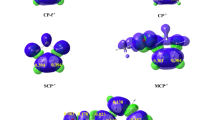
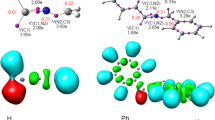
Structure effects of various alkylcyclohexenes in heterogeneously catalyzed reactions are discussed. Experimentally evaluated adsorptivity and reactivity were interpreted by means of molecular modelling. Trends in these parameters are correlated with the properties of frontier orbitals.
Similar content being viewed by others
REFERENCES
L. CervenÝ, V. Ruzicka: Adv. Catal., 30, 335 (1981).
L. CervenÝ, V. Ruzicka: Catal. Rev.-Sci. Eng., 24, 503 (1982).
M. Kraus: Adv. Catal., 29, 191 (1980).
M. Kraus, G. Ertl (Ed.), H. KnÖzinger (Ed.), J. Weitkamp (Ed.): Handbook of Heterogeneous Catalysis, p. 1051, VCH Verlagsgesellschaft mbH, Weinheim 1997.
P. Kacer, L. Låte, L. Cervenz: Collect. Czech. Chem. Commun., 63, 1915 (1998).
P. Sautet: Top. Catal., 13, 213 (2000).
J.J.P. Stewart: J. Comput. Chem., 12, 320 (1991).
C.P. Rader, H.A. Smith: J. Am. Chem. Soc., 84, 1443 (1962).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kačer, P., Låte, L., Kuzma, M. et al. Structure effects of substituents located on the cyclohexene molecular skeleton in catalytic hydrogenations. Reaction Kinetics and Catalysis Letters 78, 59–64 (2003). https://doi.org/10.1023/A:1022505714681
Issue Date:
DOI: https://doi.org/10.1023/A:1022505714681