Abstract
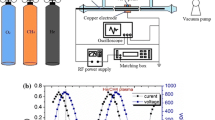
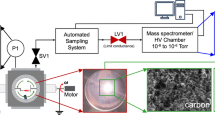
An experimental study of plasmachemical reaction involving CH4 and N2 molecules in rf discharge was studied in order to know the effect of vibrational excitation of N2 molecules. When the relative nitrogen concentration was greater than 0.8, the main product of CH4 decomposition was HCN, and the rate of methane decomposition at this condition was faster than that one in pure methane. These results could be confirmed through the mass spectroscopic method. The reason for these results is the vibrational energy of N2 excited by rf discharge. The chain reaction mechanisms of producing HCN by vibrational excitation of N2 were examined closely through numerical simulation. The rate-controlling step was the dissociation reaction of excited nitrogen molecule to the atomic nitrogen, so the process of HCN synthesis was limited by the value of reaction constant, kN.
Similar content being viewed by others
REFERENCES
V. D. Rusanov and A. A. Fridman, Physics of Chemically Active Plasma, Moscow, Nauka (1984).
S. Y. Savinov, H. Lee, H. K. Song, and B.-K. Na, Ind. Eng. Chem. Res. 38, 2540(1999).
H. Lee, S. Y. Savinov, H. K. Song, and B.-K. Na, J. Chem. Eng. Jpn. 34, 1356(2001).
Y. P. Raizer, U. N. Shneider, and N. A. Yatsenko, Radio-Frequency Capacitive Discharges, CRC Press, N.Y. (1995).
S. N. Andreev, M. A. Kerimkulov, A. A. Marurenko, V. N. Ochkin, S. Y. Savinov, and S. N. Jikhai, Tech. Phys. V. 39, 466(1994).
D. I. Slovetshii, Mechanism of Chemical Reactions in Nonequilibrium Plasma, Moscow, Nauka (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Savinov, S.Y., Lee, H., Song, H.K. et al. The Effect of Vibrational Excitation of Molecules on Plasmachemical Reactions Involving Methane and Nitrogen. Plasma Chemistry and Plasma Processing 23, 159–173 (2003). https://doi.org/10.1023/A:1022477005020
Issue Date:
DOI: https://doi.org/10.1023/A:1022477005020