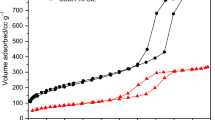
Abstract
Transmission electron microscopy, X-ray diffraction, nitrogen and argon adsorption, thermal analysis, thermoprogrammed ammonia desorption, and 1H MAS NMR spectroscopy were used to show that phosphorylation by POCl3 yields MCM-41 silica gel and Ti-MCM-41 titanium-silica gel mesoporous molecular sieves with about 1.1 mmol/g acid sites consisting largely of hydroxyl group protons of supported phosphoric acid. These materials display catalytic activity in the esterification of acetic acid by ethanol.
Similar content being viewed by others
REFERENCES
S. T. Kresge, M. E. Leonowicz, W. J. Roth, et al., Nature, 359, 710-712 (1992).
A. S. Kovalenko, V. G. Il'in, and A. P. Filippov, Teor. Éksp. Khim., 36, No. 3, 322-337 (2000).
A. S. Kovalenko, A. S. Korchev, J. V. Chernenko, et al., Adsorpt. Sci. Technol., 17, No. 4, 245-254 (1999).
O. A. Golub, O. B. Sevast'yanova, A. S. Korchev, et al., Ukr. Khim. Zh., 62, No. 12, 73-76 (1996).
B. V. Nekrasov, Fundamentals of General Chemistry [in Russian], Vol. 1, Khimiya, Moscow (1974), p. 439.
V. N. Pak, S. I. Kol'tsov, and V. B. Aleskovskii, Teor. Éksp. Khim., 12, No. 6, 839-843 (1976).
S. Imamura, S. Ishida, H. Tarumoto, et al., J. Chem. Soc., Faraday Trans., 85, No. 4, 757-762 (1993).
C. Contescu, V. T. Popa, J. M. Miller, et al., J. Catal., 157, No.1, 244-258 (1995).
M. Höctl, A. Jentys, and H. Vinek, Micropor. Mesopor. Mater., 31, No. 3, 271-285 (1999).
H. Kosslick, G. Lischke, H. Landmesser, et al., J. Catal., 176, No. 1, 102-114 (1998).
V. M. Mastikhin, I. L. Mudrakovsky, and A. V. Nosov, Progr. NMR Spectrosc., 23, 259-299 (1991).
M. H. Zahedi-Niaki, S. M. L. Laidi, and S. Kaliaguine, Micropor. Mesopor. Mater., 32, No. 3, 251-255 (1999).
S. Hitz and R. Prins, J. Catal., 168, No. 2, 194-206 (1997).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kovalenko, A.S., Korchev, A.S., Koval'chuk, T.V. et al. Effect of Phosphorylation of Si-MCM-41 and Ti-Si-MCM-41 Mesoporous Molecular Sieves on Their Structure-Adsorption and Acid Properties. Theoretical and Experimental Chemistry 38, 381–387 (2002). https://doi.org/10.1023/A:1022231905004
Issue Date:
DOI: https://doi.org/10.1023/A:1022231905004