Abstract
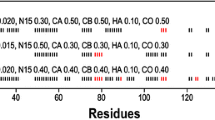
A new strategy of backbone resonance assignment is proposed based on a combination of the most sensitive TROSY-type triple resonance experiments such as TROSY-HNCA and TROSY-HNCO with a new 3D multiple-quantum HACACO experiment. The favourable relaxation properties of the multiple-quantum coherences and signal detection using the 13C′ antiphase coherences optimize the performance of the proposed experiment for application to larger proteins. In addition to the 1HN, 15N,13Cα and 13C′ chemical shifts the 3D multiple-quantum HACACO experiment provides assignment for the 1Hα resonances in constrast to previously proposed experiments for large proteins. The strategy is demonstrated with the 44 kDa uniformly 15N,13C-labeled and fractionally 35% deuterated trimeric B. subtilis Chorismate Mutase measured at 20 °C and 9 °C. Measurements at the lower temperature indicate that the new strategy can be applied to even larger proteins with molecular weights up to 80 kDa.
Similar content being viewed by others
References
Cavanagh, J., Fairbrother, W.J., Palmer, A.G. and Skelton, N.J. (1996) Protein NMR Spectroscopy: Principles and Practice. Academic Press, New York, NY.
Dayie, K.T. and Wagner, G. (1997) J. Am. Chem. Soc., 119, 7797-7806.
Eletsky, A., Kienhöfer, A. and Pervushin, K. (2001) J. Biomol. NMR, 20, 177-180.
Ernst, R.R., Bodenhausen, G. and Wokaun, A. (1987) The Principles of Nuclear Magnetic Resonance in One and Two Dimensions. Clarendon Press, Oxford.
Geen, H. and Freeman, R. (1991) J. Magn. Reson., 93, 93-141.
Grzesiek, S., Kuboniwa, H., Hinck, A.P. and Bax, A. (1995) J. Am. Chem. Soc., 117, 5312-5315.
Koehl, P. (1999) Prog. Nucl. Mag. Reson. Spectrosc., 34, 257-299.
Konrat, R., Yang, D.W. and Kay, L.E. (1999) J. Biomol. NMR, 15, 309-313.
Koradi, R., Billeter, M. and Wüthrich, K. (1996) J. Mol. Graph., 14, 51-55.
Lipari, G. and Szabo, A. (1982) J. Am. Chem. Soc., 104, 4546-4559.
Marion, D., Ikura, M., Tschudin, R. and Bax, A. (1989) J. Magn. Reson., 85, 393-399.
Mulder, F.A.A., Ayed, A., Yang, D.W., Arrowsmith, C.H. and Kay, L.E. (2000) J. Biomol. NMR, 18, 173-176.
Pervushin, K.V. (2000) Quart. Rev. Biophys., 33, 161-197.
Pervushin, K., Riek, R., Wider, G. and Wüthrich, K. (1997) Proc. Natl. Acad. Sci. USA, 94, 12366-12371.
Pervushin, K., Vögeli, B. and Eletsky, A. (2002) J. Am. Chem. Soc., 124, 12898-12902.
Salzmann, M., Pervushin, K., Wider, G., Senn, H. and Wüthrich, K. (1998) Proc. Natl. Acad. Sci. USA, 95, 13585-13590.
Salzmann, M., Pervushin, K., Wider, G., Senn, H. and Wüthrich, K. (1999a) J. Biomol. NMR, 14, 85-88.
Salzmann, M., Wider, G., Pervushin, K., Senn, H. and Wüthrich, K. (1999b) J. Am. Chem. Soc., 121, 844-848.
Salzmann, M., Wider, G., Pervushin, K. and Wüthrich, K. (1999c) J. Biomol. NMR, 15, 181-184.
Salzmann, M., Pervushin, K., Wider, G., Senn, H. and Wüthrich, K. (2000) J. Am. Chem. Soc., 122, 7543-7548.
Serber, Z., Richter, C. and Dotsch, V. (2001) Chembiochem, 2, 247-251.
Serber, Z., Richter, C., Moskau, D., Bohlen, J.M., Gerfin, T., Marek, D., Haberli, M., Baselgia, L., Laukien, F., Stern, A.S., Hoch, J.C. and Dotsch, V. (2000) J. Am. Chem. Soc., 122, 3554-3555.
Shaka, A.J., Keeler, J., Frenkiel, T. and Freeman, R. (1983) J. Magn. Reson., 52, 335-338.
Swapna, G.V.T., Rios, C.B., Shang, Z.G. and Montelione, G.T. (1997) J. Biomol. NMR, 9, 105-111.
Veeman, W.S. (1984) Prog. NMR Spectrosc., 16, 193-235.
Wüthrich, K. (1986) NMR of Proteins and Nucleic Acids, Wiley, New York, NY.
Xia, Y.L., Kong, X.M., Smith, D.K., Liu, Y., Man, D. and Zhu, G. (2000) J. Magn. Reson., 143, 407-410.
Yang, D.W. and Kay, L.E. (1999) J. Am. Chem. Soc., 121, 2571-2575.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pervushin, K., Eletsky, A. A new strategy for backbone resonance assignment in large proteins using a MQ-HACACO experiment. J Biomol NMR 25, 147–152 (2003). https://doi.org/10.1023/A:1022225711122
Issue Date:
DOI: https://doi.org/10.1023/A:1022225711122