Abstract
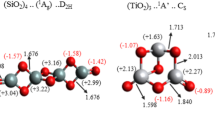
Designing principles for forming stable metallic clusters whose chemistry mimics different atoms of the periodic table are discussed. It is shown that while bulk Al is a metal, the chemistry of an Al13 resembles that of a halogen atom, a CAl12 resembles an inert atom, while a NAl12 resembles an alkali atom. The feasibility of making new materials using clusters as the building blocks is discussed. An ionic solid made out of Al13 (or BAl12) and Cs is shown to be metastable and marked by a large gap at the Fermi energy even though bulk Al and Cs are metals.
Similar content being viewed by others
REFERENCES
A. Goldstein (ed.), Handbook of Nanophase Materials (Marcel Dekker, New York, 1997).
P. Jena, S. N. Khanna, and B. K. Rao (eds.), Physics and Chemistry of Finite Systems: From Clusters to Crystals (Kluwer Academic, Dordrecht, The Netherlands, 1992).
S. N. Khanna and P. Jena (1995). Phys. Rev. 51, 13705.
W. Kratschmer, L. D. Lamb, K. Fostiropoulos, and D. R. Huffman (1990). Nature (London) 347, 354; H. W. Kroto, J. R. Heath, S. C. O'Brien, R. F. Curl, and R. E. Smalley (1985). Nature (London) 318, 162.
A. F. Hebard, M. J. Rosseinsky, R. C. Haddon, D. W. Murphy, S. H. Glarum, T. T. M. Palstra, A. P. Ramirez, and A. R. Kortan (1991). Nature 350, 600.
B. C. Guo, K. P. Kerns, and A. W. Castleman, Jr. (1992). Science 255, 1411.
S. E. Kooi and A. W. Castleman, Jr. (1998). J. Chem. Phys. 108, 8864.
W. D. Knight, K. Clementer, W. A. de Heer, W. A. Saunders, M. Y. Chou, and M. L. Cohen (1984). Phys. Rev. Lett. 52, 2141.
R. E. Leuchtner, A. C. Harms, and A. W. Castleman, Jr. (1989). J. Chem. Phys. 91, 2753.
T. P. Martin, U. Naher, and H. Schaber (1992). Chem. Phys. Lett. 199, 470.
T. P. Martin, T. Bergmann, H. Göhlich, and T. Lange (1990). Chem. Phys. Lett. 172, 209.
H. P. Cheng, R. S. Berry, and R. L. Whetten (1991). Phys. Rev. B 43, 10647; J. Y. Yi, D. J. Oh, J. Bernholc, and R. Car (1990). Chem. Phys. Lett. 174, 461; X. G. Gong and V. Kumar (1993). Phys. Rev. Lett. 70, 2078.
M. J. Frisch, G. W. Trucks, H. B. Schegel, P. M. W. Gill, B. G. Johnson, M. A. Robb, J. R. Cheeseman, T. Keith, G. A. Petersson, J. A. Montgomery, K. Raghavachari, M. A. Al-Laham, V. G. Zakrzewski, J. V. Ortiz, J. B. Foresman, J. Closlowski, B. B. Stefanov, A. Nanayakkara, M. Challacombe, C. Y. Peng, P. Y. Ayala, W. Chen, M. W. Wong, J. L. Andres, E. S. Replogle, R. Gomperts, R. L. Martin, D. J. Fox, J. S. Binkley, D. J. Defrees, J. Baker, J. P. Stewart, M. Heads-Gordon, C. Gonzalez, and J. A. Pople, Gaussian 94, rev. B.1 (Gaussian, Inc., Pittsburgh, PA, 1995).
M. F. Jarrold, J. E. Bower, and S. J. Kraus (1987). J. Chem. Phys. 86, 3876.
S. N. Khanna and P. Jena (1992). Phys. Rev. Lett. 69, 1664.
A. P. Seitsonen, M. J. Puska, M. Alatalo, R. M. Nieminen, V. Milman, and M. C. Payne (1993). Phys. Rev. B 48, 1981.
R. Kawai, private communication.
X. G. Gong (1997). Phys. Rev. B 56, 1091.
S. N. Khanna and P. Jena (1994). Chem. Phys. Lett. 218, 383.
S. N. Khanna and P. Jena (1994). Chem. Phys. Lett. 219, 479.
S. K. Nayak, S. N. Khanna, and P. Jena (1998). Phys. Rev. B 57, 3787.
F. Liu, M. Mostoller, T. Kaplan, S. N. Khanna, and P. Jena (1996). Chem. Phys. Lett. 248, 213.
C. Ashman, S. N. Khanna, F. Liu, P. Jena, T. Kaplan, and M. Mostoller (1997). Phys. Rev. B 55, 15868.
F. Kirchoff, X. G. Gong, J. W. Wilkins, F. S. Khan, G. Kresse, and J. Hafner (1998). Bull. Am. Phys. Soc. 43, 28.
S. C. Sevov and J. D. Corbett (1993). Science 262, 880.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rao, B.K., Khanna, S.N. & Jena, P. Designing New Materials Using Atomic Clusters. Journal of Cluster Science 10, 477–491 (1999). https://doi.org/10.1023/A:1021948806958
Issue Date:
DOI: https://doi.org/10.1023/A:1021948806958