Abstract
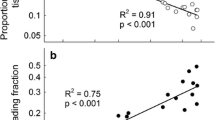
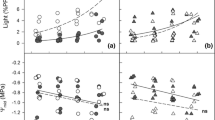
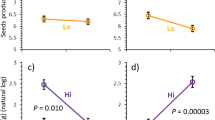
The carbon–nutrient balance hypothesis (CNBH) predicts that shading should increase leaf palatability to herbivores by decreasing concentrations of carbon (C) -based chemical defenses and increasing nitrogen (N). We measured cottonwood (Populus deltoids) growth, leaf chemistry, and beetle (Plagiodera versicolora) feeding preferences on saplings grown in either continuous high (HH) or low (LL) light, and saplings switched from high to low (HL) or low to high (LH) light for nine days. As expected, based on the CNBH, shading increased total N and decreased total phenol glycoside (C-based secondary metabolites) concentrations in plants from all shade treatments (LL, HL, and LH), relative to HH plants, with plant growth and gross leaf chemistry being affected by initial and final light regime. In contrast, while specific phenol glycoside concentrations were affected by the initial and final light regime, they also showed an initial × final light interaction. Beetles tended to prefer LL to HH plants. Beetles unexpectedly preferred HH to either HL or LH switched plants, most likely because high concentrations of a specific phenol glycoside – salicin – occurred in both switched treatments and inhibited beetle feeding. Plant chemical allocation during light acclimation led to unpredictable changes in specific C-based compounds, even though plant growth and gross chemistry conformed to expectations for shading effects and the CNBH. The response of this herbivore to altered concentrations of a specific compound confounded predictions based on average dynamics of suites of chemicals. Our findings may help explain why relationships between light availability and herbivory in field studies, where light varies on many time scales, can differ from those predicted by the CNBH. Understanding both dynamic plant chemical responses to altered resource availability and controls over allocation to specific compounds would likely enhance future predictability of specific environment-plant-herbivore interactions.
Similar content being viewed by others
REFERENCES
Aide, T. M., and Zimmerman, J. K. 1990. Patterns of insect herbivory, growth, and survivorship in juveniles of a neotropical liana. Ecology 41:1412-1421.
Barker, H. R., and Barker, B. M. 1984. Multivariate Analysis of Variance (MANOVA). The University of Alabama Press, University, Alabama.
Basset, Y. 1991. The spatial distribution of herbivory, mines and galls within an Australian rain forest tree. Biotropica 23:271-281.
Bingaman, B. R., and Hart, E. R. 1993. Clonal and leaf age variation in Populus phenolic glycosides: Implications for host selection by Chrysomela scripta (Coleoptera: Chrysomelidae). Environ. Entomol. 22:397-403.
Bryant, J. P. 1987. Feltleaf willow-snowshoe hare interactions: Plant carbon/nutrient balance and floodplain succession. Ecology 68:1319-1327.
Bryant, J. P., Chapin, F. S., and Klein, D. R. 1983. Carbon/nutrient balance of boreal plants in relation to vertebrate herbivory. Oikos 40:357-368.
Bryant, J. P., Chapin, F. S., Reichardt, P. B., and Clausen, T. P. 1987. Response of winter chemical defense in Alaska paper birch and green alder to manipulation of plant carbon/nutrient balance. Oecologia 72:510-514.
Coleman, J. S., and Jones, C. G. 1988. Plant stress and insect performance: Cottonwood, ozone and a leaf beetle. Oecologia 76:57-61.
Collinge, S. K., and Louda, S. M. 1988. Herbivory by leaf minors in response to experimental shading of a native crucifer. Oecologia 76:559-566.
Dudt, J. F., and Shure, D. J. 1994. The influence of light and nutrients on foliar phenolics and herbivory. Ecology 75:86-89.
Folgarait, P. J., Marquis, R. J., Ingvarsson, P., Braker, H. E., and Arguedas, M. 1995. Patterns of attack by insect herbivores and a fungus on saplings in a tropical tree plantation. Environ. Entomol. 24:1487-1494.
Grime, J. P. 1966. Shade avoidance and shade tolerance in flowering plants, pp. 187-207, in R. Bainbridge, G. C. Evans, and O. Rackham (eds.). Light as an Ecological Factor. Blackwell Scientific, Oxford.
Harrison, S. 1987. Treefall gaps versus forest understory as environments for a defoliating moth on a tropical forest shrub. Oecologia 72:65-68.
Hosner, J. F., and Minckler, L. F. 1963. Bottomland and hardwood forests of southern Illinois—regeneration and succession. Ecology 44:29-41.
Jones, C. G., and Coleman, J. S. 1988. Plant stress and insect behavior: Cottonwood, ozone, and the feeding and oviposition preference of a beetle. Oecologia 76:51-56.
Jones, C. G., and Coleman, J. S. 1989. Biochemical indicators of air pollution in trees: Unambiguous signals based on secondary metabolites and nitrogen in fast-growing species, pp. 261-273, in National Research Council (eds.). Biologic Markers Air Pollution Stress and Damage in Forests. National Academy Press, Washington, D.C.
Jones, C. G., Hare, J. D., and Compton, S. 1989. Measuring plant protein with the Bradford assay. J. Chem. Ecol. 15:979-992.
Jones, C. G., Hopper, R. F., Coleman, J. S., and Krishik, V. A. 1993. Control of systemically induced herbivore resistance by plant vascular architecture. Oecologia 93:452-456.
Jones, C. G., Coleman, J. S., and Findlay, S. 1994. Effects of ozone on interactions between plants, consumers and decomposers, pp. 339-363, in R. G. Alsher and A. Wellburn (eds.). Plant Responses to the Gaseous Environment. Chapman and Hall, London.
Larcher, W. 1980. Plant Physiological Ecology. Springer-Verlag, New York.
Larson, P. R., and Isebrands, J. G. 1971. The plastochron index as applied to developmental studies of cottonwood. Can. J. For. Res. 1:1-11.
Larsson, S., Wiren, A., Lundgren, L., and Ericsson, T. 1986. Effects of light and nutrient stress on leaf phenolic chemistry in Salix dasclados and susceptibility to Galerucella lineola. Oikos 47:205-210.
Lincoln, D. E., and Mooney, H. A. 1984. Herbivory on Diplacus aurantiacus shrubs in sun and shade. Oecologia 64:173-176.
Lindroth, R. L., and Pajutee, M. S. 1987. Chemical analysis of phenolic glycosides: Art, facts, and artifacts. Oecologia 74:144-148.
Lindroth, R. L., Hsia, M. T. S., and Scriber, J. M. 1987. Seasonal patterns on the phytochemistry of three Populus species. Biochem. Syst. Ecol. 15:681-686.
Lindroth, R. L., Reich, P. B., Tjoelker, M. G., Volin, J. C., and Oleksyn, J. 1993. Light environment alters response to ozone stress in seedlings of Acer saccharum Marsh. and hybrid Populus L. III. Consequences for performance of gypsy moth. New Phytol. 124:647-651.
Louda, S. M., and Rodman, J. E. 1983. Concentration of glucosinolates in relation to habitat and insect herbivory for the native crucifer Cardamine cordifolia. Biochem. Syst. Ecol. 11:199-207.
Louda, S. M., Dixon, P. M., and Huntly, N. J. 1987. Herbivory in sun versus shade at a natural meadow-woodland ecotone in the Rocky Mountains. Vegetatio 72:141-149.
Lowman, M. D. 1992. Leaf growth dynamics and herbivory in five species of Australian rain-forest canopy tree. J. Ecol. 80:433-447.
Maiorana, V. C. 1981. Herbivory in sun and shade. Biol. J. Linn. Soc. 15:151-156.
Mole, S., and Waterman, P. G. 1988. Light-induced variation in phenolic levels in foliage of rainforest plants. II. Potential significance to herbivores. J. Chem. Ecol. 14:23-34.
Mole, S., Ross, J. A. M., and Waterman, P. G. 1988. Light-induced variation in phenolic levels in foliage of rainforest plants. J. Chem. Ecol. 14:1-21.
Nichols-Orians, C. 1991. Environmentally induced differences in plant traits: Consequences for susceptibility to a leaf-cutter ant. Ecology 72:1609-1623.
Orians, C. M. 1995. Preserving leaves for tannin and phenolic glycoside analysis: A comparison of methods using three willow taxa. J. Chem. Ecol. 21:1235-1243.
Peterson, N. C., and Smitley, D. R. 1991. Susceptibility of selected shade and flowering trees to gypsy moth (Lepidoptera: Lymantridae). J. Econ. Entomol. 84:587-592.
Picard, S., Chenault, J., Augustin, S., and Venot, C. 1994. Isolation of a new phenolic compound from leaves of Populus deltoides. J. Nat. Prod. 57:808-810.
Reichardt, P. B., Chapin, F. S., Bryant, J. P., Mattes, B. R., and Clausen, T. P. 1991. Carbon/nutrient balance as a predictor of plant defense in Alaskan balsam poplar: Potential importance of metabolite turnover. Oecologia 88:401-406.
Scheiner, S. M. 1993. MANOVA: Multiple response variables and multispecies interactions, pp. 94-112, in S. M. Scheiner and J. Gurevitch (eds.). Design and Analysis of Ecological Experiments. Chapman and Hall, New York.
Sims, D. A., and Pearcy, R. W. 1991. Photosynthesis and respiration in Alocasia macrorriza following transfers to high and low light. Oecologia 86:447-453.
Smith, H. 1981. Adaptation to shade, pp. 159-173, in C. B. Johnson (ed.). Physiological Processes Limiting Plant Productivity. Butterworths, London.
Tahvanainen, J., Helle, E., and Kettunen, J. 1985. Phenolic levels govern the food selection pattern of willow feeding leaf beetles. Oecologia 67:52-56.
Wade, M. J., and Breden, F. 1986. Life history of natural populations of the imported willow leaf beetle, Plagiodera versicolora (Coleoptera: Chrysomelidae). Ann. Entomol. Soc. Am. 55:171-180.
Wait, D. A. 1992. Effect of nutrient addition rates on growth, chemistry, and physiology of Populus deltoides, and the feeding preference and consumption of Chrysomela scripta and Lymantria dispar. MS thesis. State University of New York, College of Environmental Science and Forestry, Syracuse, New York.
Wait, D. A., Jones, C. G., and Coleman, J. S. 1998. Effects of nitrogen fertilization on leaf chemistry and beetle feeding are mediated by leaf development. Oikos. 82:502-504.
Waring, R. H., McDonald, A. J. S., Larsson, S., Ericsson, T., Wiren, A., Arwidsso, E., Ericsson, A., and Lohammar, T. 1985. Differences in chemical composition of plants grown at constant relative growth rates with stable mineral nutrition. Oecologia 66:157-160.
Waterman, P. G., Ross, J. A. M., and McKey, D. B. 1984. Factors affecting levels of some phenolic compounds, digestibility, and nitrogen content of the mature leaves of Barteria fistulosa (Passifloraceae). J. Chem. Ecol. 10:387-401.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Crone, E.E., Jones, C.G. The Dynamics of Carbon–Nutrient Balance: Effects of Cottonwood Acclimation to Short- and Long-Term Shade on Beetle Feeding Preferences. J Chem Ecol 25, 635–656 (1999). https://doi.org/10.1023/A:1020966206840
Issue Date:
DOI: https://doi.org/10.1023/A:1020966206840