Abstract
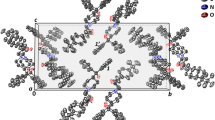
The X-ray crystal structures of three ergoline derivatives have been completed and are reported herein. These include mesulergine hydrochloride (III), pergolide methanesulfonate (IV), and dihydroergocriptine hydrate (V). These molecules show dopamine agonist activity at the D2 receptor. These three-dimensional structures were compared to structurally similar molecules with similar pharmacological activity. It was found that ergoline derivatives have rigid fused-ring systems with substituents that can exist in a variety of conformations. The ergoline structures reported here have structural features consistent with other active dopamine agonists. Crystallographic data: (III) C18H26N4O2S ⋅ HCl, monoclinic space group P21, a = 12.183(2) Å, b = 6.174(2) Å, c = 13.649(2) Å, β = 105.55(1)°, Z = 2, final R = 0.071 for 2293 observed reflections [I > 3σ(I)]; (IV) C19H27N2S ⋅ CH3SO3, mono- clinic space group P21, a = 8.455(4) Å, b = 29.868(4) Å, c = 8.957(6) Å, β = 111.95(5)°, Z = 4, final R = 0.055 for 3836 observed reflections; (V) C32H43N5O5 ⋅ H2O, monoclinic space group P21, a = 12.403(2) Å, b = 18.103(3) Å, c = 15.418(3) Å, β = 113.04(2)°, Z = 4, final R = 0.058 for 3208 observed reflections.
Similar content being viewed by others
REFERENCES
Arky, R. Physicians' Desk Reference; Medical Economics Data Production Co.: Montvale, NJ: 1998.
Liang, L.; Zhu, N.; White, J.; Brown, C.; Klein Stevens, C. L. J. Pharm. Sci. 1998,87(12), 1496-1501.
Klein, C. L.; Lear, J.; O'Rourke, S.; Williams, S.; Liang, L. J. Pharm. Sci. 1994,83(9), 1253-1256.
Piccoli, F.; Riuggeri, R. M. J. Neural Transm. 1995(Suppl.), 45, 187-195.
Lieberman, A. N.; Neophytides, A.; Leibowitz, M. Advan. Neurol. 1983,45, 95-198.
Battino, M.; Littarru, G. P.; Gorini, A.; Villa, R. F. Neurochem. Res. 1996,21(12), 1505-1514.
MolEN, Structure Determination System of programs used for the processing, solving, and interpretation of the data was written by C. K. Fair, 1990.
Main, P.; Fiske, S. J.; Hull, S. E.; Lessinger, L.; Germain, G.; Declercq, J.-P.; Woolfson, M. M. MULTAN11/82. A System of Computer Programs for the Automatic Solution of Crystal Structures from X-ray Diffraction Data; Universities of York, England and Louvain, Belgium.
International Tables for X-ray Crystallography; Kynoch Press: Birmingham, England, 1974; Vol. IV, pp. 71-151.
Ma, L. Y. Y.; Camerman, N.; Swartzendruber, J. K.; Jones, N. D. Can. J. Chem. 1987,65, 256-260.
Camerman, N.; Chan, L. Y. Y.; Camerman, A. Mol. Pharmacol. 1979,16, 729-736.
Čejka, J.; Horák, Z.; Pakhomova, S.; Kratochvíl, B.; Sedmera, P.; Havíček, V.; Cvak, L.; Jegorov, A. Collect. Czech. Chem. Commun. 1998,63, 803-812.
Bergin, R.; Carlstrom, D. Acta Crystallogr. 1968,B24, 1506-1510.
Seeman, P. Pharmacol. Rev. 1981,32, 229-313.
Tonani, R,; Dunbar, J.; Edmonston, B.; Marshall, G. R. J. Comp. Aid. Mol. Design 1987,1, 121-132.
Wikstrom, H.; Andersson, B.; Sanchez, D.; Lindberg, P.; Arvidsson, L.-E.; Johansson, A. M.; Nilsson, J. L. G.; Svensson, K.; Hjorth, S.; Carlsson, A. J. Med. Chem. 1985,28, 215-225.
Seeman, P.; Watanabe, M.; Grigoriadis, D.; Tedesco, J. L.; George, S. R.; Svensson, U.; Nilsson, J. L. G.; Neumeyer, J. L. Mol. Pharmacol. 1985,28, 391-399.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhu, N., Johnson, L., White, J. et al. A Crystallographic Investigation of Ergolines. Structural Chemistry 13, 491–499 (2002). https://doi.org/10.1023/A:1020521706309
Issue Date:
DOI: https://doi.org/10.1023/A:1020521706309