Abstract
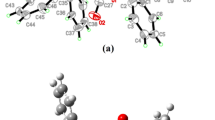
The crystal structure of hydrazinium(2+) aquatetrafluoroindate(III), N2H6(InF4H2O)2, has been determined by X-ray diffraction. The compound crystallizes in the triclinic space group \(P\bar 1\) (C i 1) with a = 6.759(2) Å, b = 6.228(1) Å, c = 5.7854(7) Å, α = 80.28(2)°, β = 88.79(1)°, γ = 69.96(2)°, V = 225.3(1) Å3, and Z = 1. The structure comprises hydrazinium(2+) cations and InF2F4/2O− anions in which In appears to be seven-coordinated in the form of a pentagonal bipyramid by four bridging fluorine atoms, two terminal fluorine atoms, and one oxygen atom. Pentagonal bipyramids are interconnected and form chains along a axis. Bands in vibrational spectra were assigned to the vibrations in N2H6 2+ cations and anionic InF2F4/2O− entities. The rather intensive band appearing at 1042 cm−1 in Raman spectrum, which is absent from infrared, is characteristic of the N2H6 2+ cation, in line with the predictions of the unit-cell group analysis for the studied system.
Similar content being viewed by others
References
Šiftar, J.; Bukovec, P. Monatsh. Chem. 1970, 101, 1184.
Bukovec, P.; Šiftar, J. Monatsh. Chem. 1971, 102, 94.
Keppert, D.L. Prog. Inorg. Chem. 1979, 25, 41.
Christe, O.K.; Sanders, J.C.P.; Schrobilgen, G.J.; Wilson, W.W. J. Chem. Soc., Chem. Commun. 1991, 13, 837.
Christe, K.O.; Curtis, E.C.; Dixon, D.A.; Mercier, H.P.A.; Sanders, J.C.P.; Schrobilgen, G.J.; Wilson, W.W. In Inorganic Fluorine Chemistry: Toward the 21st Century: Heptacoordinated Main-Group Fluorides and Oxofluorides; Thrasher, J.S.; Strauss, S.H., Eds.; ACS Symposium Series 555; ACS: Washington, DC, 1994; pp 66–89.
Drake, G.W.; Dixon, D.A.; Sheehy, J.A.; Boatz, J.A.; Christe, K.O. J. Am. Chem. Soc. 1998, 120, 8392.
Seppelt, K. In Inorganic Fluorine Chemistry: Toward the 21st Century: Main-Group Fluorides with Coordination Numbers Greater-than-Six: Thrasher, J.S.; Strauss, S.H., Eds.; ACS Symposium Series 555; ACS: Washington, DC, 1994; pp 56–65.
Wells, A.F. Structural Inorganic Chemistry; Clarendon Press: Oxford, 1984; pp 471–474.
Bukovec, P.; Goliě, L. Acta Crystallogr. Sect. B, 1976, 32, 948.
Fisher, P.J.; Weiss, R. Acta Crystallogr., Sect. B 1973, 29, 1958.
Laval, P.J.P.; Papiernik, R.; Frit, B. Acta Crystallogr., Sect. B 1978, 34, 1070.
Hurst, H.J.; Taylor, J.C. Acta Crystallogr., Sect. B 1970, 26, 2136.
Keller, O.L. Inorg. Chem. 1963, 2(4), 783; Pausewang, V.G.; Schmitt, R.; Dehnicke, K. Z. Anorg. Allg. Chem., 1974, 408, 1.
Poojary, M.D.; Patil, K.C. Proc. Indian Acad. Sci. (Chem. Sci.) 1987, 99, 311.
Almond, P.M.; Talley, C.E.; Bean, A.C.; Peper, S.M.; Albrecht-Schmitt, T.E. J. Solid State Chem. 2000, 154, 635.
Zachariasen, W.A. Acta Crystallogr. 1954, 7, 783; Acta Crystallogr. 1954, 7, 792.
Benkiê P.; Rahten A.; Jesih A. Posvetovanja Slov. Kem. Dnevi 2000, (Pt. 1), 221.
McBride, W.R.; Henry, R.A.; Skolnik, S. Anal. Chem. 1951, 23, 890.
Sedej, B. Talanta 1976, 23, 335.
Kovington, A.K. Ion-Selective Electrode Methodology; CRC Press: Florida, 1984; Vol. II, pp 76–80.
Pribil, R. Applied Complexometry; Pergamon Press: Oxford, 1982; Vol. 5; pp 121–122.
teXsan for Windows version 1.06; Crystal Structure Analysis Package: Molecular Structure Corp., 1999.
Becke, A.D. J. Chem. Phys. 1993, 98, 5648.
Lee, C.; Yang, W.; Parr, R.G. Phys. Rev. B1988, 37, 785.
Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Zakrzewski, W.G.; Montgomery, J.A.; Stratmann, R.E.; Burant, J.C.; Dapprich, S.; Millam, J.M.; Daniels, A.D.; Kudin, K.N.; Strain, M.C.; Farkas, O.; Tomasi, J.; Barone, V.; Cossi, M.; Cammi, R.; Mennucci, R.; Pomelli, C.; Adamo, C.; Clifford, S.; Ochterski, J.; Petersson, G.A.; Ayala, P.Y.; Cui, Q.; Morokuma, K.; Malick, D.K.; Rabuck, A.D.; Raghavachari, K.; Foresman, J.B.; Cioslowski, J.; Ortiz, J.V.; Stefanov, B.B.; Liu, G.; Liashenko, A.; Piskorz, P.; Komaromi, I.; Gomperts, R.; Martin, R.L.; Fox, D.J.; Keith, T.; Al-Laham, M.A.; Peng, C.Y.; Nanayakkara, A.; Gonzalez, C.; Challacombe, M.; Gill, P.M.W.; Johnson, B.G.; Chen, W.; Wong, M.W.; Andres, J.L.; Head-Gordon, M.; Replogle, E.S.; Pople, J.A. Gaussian98, Revision A.9; Gaussian, Inc.: Pittsburgh, PA, 1998.
Cameron, T.S.; Knop, O.; Macdonald, L.A. Can. J. Chem. 1983, 61, 184.
Gillespie, R.J. Molecular Geometry; Van Nostrand Reinhold Co.: London, 1972.
Gillespie, R.J.; Hargittai I. The VSEPR Model of Molecular Geometry; Allyn and Bacon, A Division of Simon & Schuster, Inc.: Needham Heights, MA, 1991.
Christe, K.O.; Dixon, D.A.; Sanders, C.P.; Schrobilgen, G.J.; Wilson, W.W. J. Am. Chem. Soc. 1993, 115, 9461.
Christe, K.O.; Dixon, D.A.; Sanders, C.P.; Schrobilgen, G.J.; Tsai, S.S.; Wilson, W.W. Inorg. Chem. 1995, 34, 1868.
Reisfeld, M.J. Spectrochim. Acta, Part A 1973, 29, 1923.
Bukovec, P.; Orel, B.; Šiftar, J. Monatsh. Chem. 1971, 102, 885.
Keller, O.L., Jr.; Chetham-Strode, A., Jr. Inorg. Chem. 1966, 5(3), 367.
Pejov, Lj.; Petruševski, V.M.; Rahten, A.; Jesih, A.; Skapin, T. J. Mol. Struct. 2002, 604, 1.
Milićev, S.; Rahten, A.; Borrmann, H.; Šiftar, J. J. Raman Spectrosc. 1997, 28, 315.
MiliŰev, S.; Maček, J. Spectrochim. Acta Part A, 1985, 41, 55.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Benkič, P., Rahten, A., Jesih, A. et al. Crystal structure and vibrational spectra of hydrazinium(2+) aquatetrafluoroindate(III), N2H6(InF4H2O)2 . Journal of Chemical Crystallography 32, 227–235 (2002). https://doi.org/10.1023/A:1020244905386
Issue Date:
DOI: https://doi.org/10.1023/A:1020244905386