Abstract
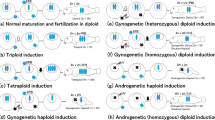
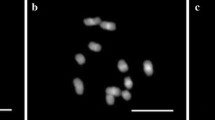
Polyploid gibel carp, Carassius auratus gibelio, is an excellent model system for evolutionary genetics owing to its specific genetic background and reproductive modes. Comparative karyotype studies were performed in three cultured clones, one artificially manipulated group, and one mated group between two clones. Both the clones A and P had 156 chromosomes in their karyotypes, with 36 metacentric, 54 submetacentric, 36 subtelocentric, 24 acrocentric, and six small chromosomes. The karyotype of clone D contained 162 chromosomes, with 42 metacentric, 54 submetacentric, 36 subtelocentric, 24 acrocentric, and six small chromosomes. All the three clones had six small chromosomes in common. Group G, being originated from the clone D by artificial manipulation, showed supernumerary microchromosomes or chromosomal fragments, in addition to the normal chromosome complement that was identical to the clone D. The offspring from mating between clones D and A had 159 chromosomes. Comparing with the clone A, the DA offspring showed three extra metacentric chromosomes. In addition, variable RAPD fingerprint patterns and unusual SCAR marker inheritance were, respectively, detected among individuals of artificial group G and in the mated DA offspring. Both the chromosome and molecular findings suggest that genome reshuffling might have occurred by manipulation or mating of the clones.
Similar content being viewed by others
References
Amores, A., A. Force, Y.L. Yan, L. Joly, C. Amemiya, A. Fritz, R.K. Ho, J. Langeland, V. Prince, Y.L. Wang, M. Westerfield, M. Ekker &; J.H. Postlethwait, 1998. Zebrafish Hox clusters and vertebrate genome evolution. Science 282: 1711–1714.
Beçak, M.L. &; W. Beçak, 1974a. Diploidization in Eleutherodactylus (Leptodactylidae-Amphibia). Experimentia 30: 624–625.
Beçak, M.L. &; W. Beçak, 1974b. Studies on polyploid amphibians: karyotype evolution and phylogeny of the genus Odontophrynus. J. Herpet. 8: 337–341.
Fan, Z. &; J. Shen, 1990. Studies on the evolution of bisexual reproduction in silver crucian carp (Carassius auratus gibelio Bloch). Aquaculture 84: 235–244.
Fan, L.C., J.F. Gui, J. Ding, L.F. Zhu, S.C. Liang &; Z.A. Yang, 1997. Cytological mechanism on the integration of heterologous genome or chromosomes in the unique gynogenetic Carassius auratus gibelio. Develop. Reproduct. Biol. 6: 33–44.
Fan, L.C., Y.P. Lai, L.F. Zhu, S.C. Liang &; J.F. Gui, 2000. Comparative studies on mitochondrial DNAs of two different clones of Carassius auratus gibelio. Oceanol. Limnol. Sinica 31(4): 370–377.
Fan, L.C., S.T. Yang &; J.F. Gui, 2001. Differential screening and characterization analysis of the egg envelope glycoprotein ZP3 cDNAs between gynogenetic and gonochoristic crucian carp. Cell Res. 11(1): 17–27.
Gui, J.F., 1989. Evolutionary genetic of unisexual vertebrates. Nature J. 12: 116–122.
Gui, J.F., 1996. A unique study system: gynogenetic fish Carassius auratus gibelio. Sci. Found. China 4: 44–46.
Gui, J.F., 1999. Fish developmental genetics and artificial propagation, pp. 41–62 in Fish Genetics and Breeding Engineering, edited by C. Wu &; J.F. Gui. Shanghai Scientific and Technical Publishers, Shanghai.
Gui, J.F., S.C. Liang &; J.M. Sun, 1990. Studies on genome manipulation in fish. I. Induction of triploid transparent colored crucian carp by hydrostatic pressure. Acta Hydrobiol. Sinica 14(4): 336–344.
Gui, J.F., S.C. Liang, L.F. Zhu &; Y.G. Jiang, 1993a. Discovery of multiple tetraploids in artificially propagated populations of allogynogenetic silver crucian carp and their breeding potentialities. Chinese Sci. Bull. 38(4): 327–331.
Gui, J.F., S.C. Liang, L.F. Zhu &; Y.G. Jiang, 1993b. Discovery of two different reproductive development modes of the eggs of artificial multiple tetraploid allogynogenetic silver crucian carp. Chinese Sci. Bull. 38(4): 332–337.
Jiang, Y.G., H.X. Yu, B.D. Chen, S.C. Liang, 1983. Biological effect of heterologous sperm on gynogenetic offspring in Carassius auratus gibelio. Acta Hydrobiol. Sinica 8: 1–13.
Kobayashi, H., K. Nakano &; M. Nakamura, 1977. On the hybrids, 4n ginbuna (C. auratus langsdorfii × kinbuna (C. auratus subsp.) and their chromosome. Bull. Jpn. Soc. Sci. Fish. 43: 31–37.
Kondrashov, A.S., 1994. The asexual ploidy cycle and the origin of sex. Nature 370: 213–216.
Levan, A., K. Fredge &; A.A. Sandberg, 1964. Nomenclature for centromeric position of chromosomes. Hereditas 52: 210–220.
Ohno, S., J. Muramoto &; L. Christian, 1967. Diploid-tetraploid relationship among old-world members of fish family Cyprinidae. Chromosoma 23: 1–9.
Ojima, Y., S. Hitotsumachi &; S. Makino, 1966. Cytogenetic studies in lower vertebrates. I. A preliminary report on the chromosomes of the funa (Carassius auratus) and the goldfish. Proc. Jpn. Acad. 42: 62–66.
Quattro, J.M., J.C. Avise &; R.C. Vrijenhoek, 1991. Molecular evidence for multiple origins of hybridogenetic fish clones (Poeciliidae: Poeciliopsis). Genetics 127: 391–398.
Sambrook, J., E.F. Fritsch &; T. Maniatis, 1989. Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory, New York, 2nd edn, pp. 463–468.
Schartl, M., I. Nanda, I. Schlupp, B. Wilde, J.T. Epplen, M. Schmid &; J. Parzefall, 1995. Incorporation of subgenomic amounts of host species DNA in the gynogenetic Amazon Molly. Nature 373: 68–71.
Sites Jr., J.W., D.M. Peccinini-Seale, C. Moritz, J.W. Wright &; W.M. Brown, 1990. The evolutionary history of parthenogenetic Cnemidophorus lemniscatus (Sauria: Teiidae). 1. Evidence for a hybrid origin. Evolution 44: 906–921.
Soltis, D.E.&; P.E. Soltis, 1999. Polyploidy: recurrent formation and genome evolution. TREE 14: 348–352.
Vrijenhoek, R.C., 1994. Unisexual fish: model system for studying ecology and evolution. A Rev. Ecol. Syst. 25: 71–96.
Vrijenhoek, R.C., R.M. Dawley, C.J. Cole &; J.P. Bogart, 1989. A list of known unisexual vertebrates, pp. 19–23 in Evolution and Ecology of Unisexual Vertebrates, Bulletin 466, edited by R.M. Dawley &; J.P. Bogar. New York State Museum, Albany.
Yang, X.Q., M.R. Chen, X.M. Yu &; H.X. Chen, 1992. Preliminary studies on the mode of reproduction in crucian carp (Carassius auratus) of Pengze. Acta Hydrobiol. Sinica 16: 277–280.
Yang, L., S.T. Yang, X.H. Wei &; J.F. Gui, 2001. Genetic diversity among different clones of the gynogenetic silver crucian carp, Carassius auratus gibelio, revealed by transferrin and isozyme markers. Biochem. Genet. 39(5/6): 213–225.
Yu, X.J., T. Zhou, Y.C. Li, K. Li &; M. Zhou, 1989. Chromosomes of Chinese freshwater fishes. Beijing: Sci. (in press).
Zhou, L., L.C. Fan &; J.F. Gui, 1998. RAPD analysis of incorporation of heterologous genetic materials in multiple species of silver crucian carp. Acta Hydrobiol. Sinica 22(4): 301–306.
Zhou, L., Y. Wang &; J.F. Gui, 2000a. Analysis of genetic heterogeneity among five gynogenetic clones of silver crucian carp, Carassius auratus gibelio Bloch, based on detection of RAPD molecular markers. Cytogenet. Cell Genet. 88: 133–139.
Zhou, L., Y. Wang &; J.F. Gui, 2000b. Genetic evidence for gonochoristic reproduction in gynogenetic silver crucian carp (Carassius auratus gibelio Bloch) as revealed by RAPD assays. J. Mol. Evol. 51: 498–506.
Zhou, L., Y. Wang &; J.F. Gui, 2001a. Molecular analysis of silver crucian carp (Carassius auratus gibelio Bloch) clones by SCAR markers. Aquaculture 201: 219–228.
Zhou, L., Y. Wang &; J.F. Gui, 2001b. Studies on genetic diversity in gonochoristic offspring produced from mating between two different gonogenetic clones of silver crucian carp. Acta Biol. Exp. Sinica 34(3): 169–176.
Zhu, L.F. &; J.F. Gui, 1998. Comparative studies on gene expression of isozymes in the 4 types of various ploidy crucian carp during embryogenesis. Acta Biol. Exp. Sinica 31(4): 369–376.
Zhu, L.F. &; Y.G. Jiang, 1987. Intraspecific genetic markers of crucian carp (Carassius auratus gibelio) and their application to selective breeding. Acta Hydrobiol. Sinica 11(2): 105–111.
Zhu, L.F. &; Y.G. Jiang, 1993. A comparative study of the biological characters of gynogenetic clones of silver crucian carp (Carassius auratus gibelio). Acta Hydrobiol. Sinica 17(2): 112–120.
Zou, Z., Y. Cui, J. Gui &; Y. Yang, 2001. Growth and feed utilisation in two clones of gibel carp, Carassius auratus gibelio: paternal effects in a gynogenetic fish. J. Appl. Ichthyol. 17(2): 54–58.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhou, L., Gui, J. Karyotypic Diversity in Polyploid Gibel Carp, Carassius Auratus Gibelio Bloch. Genetica 115, 223–232 (2002). https://doi.org/10.1023/A:1020102409270
Issue Date:
DOI: https://doi.org/10.1023/A:1020102409270