Abstract
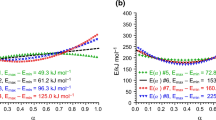
A number of 7 complexes of the [Co(DH)2(amine)2)]I type (DH2 stands for dimethyloxime) have been studied by means of thermogravimetry and differential scanning calorimetry in nitrogen atmosphere, by using heating rates of 2.5, 5 and 10 K min−1. In all cases an endothermal deamination reaction occurs leading to the relatively stable [Co(DH)2I(amine)] intermediate. For this reaction apparent kinetic parameters have been derived. The influence of heating rate is discussed. The validity of a linear and a non-linear kinetic compensation law was verified.
Similar content being viewed by others
References
C. J. Keattch, An Introduction to Thermogravimetry, Heyden and Son Ltd., London, Sadtler Research Laboratories Inc., Philadelphia 1969, p. 21.
J. Zsakó, J. Chim. Phys., 66 (1969) 1041.
J. Simon, J. Thermal Anal., 5 (1973) 271.
R. C. Turner, J. Hoffmann and D. Chen, Canadian J. Chem., 41 (1963) 243.
D. Furnica and J. A. Schneider, Makromol. Chem., 108 (1967) 182.
J. A. Schneider, C. Vasile, D. Furnica and A. Onu, Makromol. Chem., 117 (1968) 41.
C. N. Caşcaval, C. Vasile and J. A. Schneider, Makromol. Chem., 131 (1970) 55.
J. C. Hisatsune, E. C. Beahm and R. J. Kempf, J. Phys. Chem., 74 (1970) 3444.
I. G. Murgulescu, D. Fătu and E. Segal, J. Thermal Anal., 1 (1969) 97.
J. Zsakó, E. Kékedy and Cs. Várhely, Proc. 3rd ICTA, Davos 1971, p. 487.
J. Zsakó, Cs. Várhelyi and E. Kékedy, Proc. 4th ICTA, Budapest 1974, p. 177.
J. Zsakó, Cs. Várhelyi, B. Csegedi and J. Zsakó, Thermochim. Acta, 45 (1981) 11.
J. Zsakó, M. Várhelyi and Cs. Várhelyi, J. Thermal Anal., 17 (1979) 123.
J. Zsakó, Cs. Várhelyi and S. Magyarósi, Studia Univ. Babeş-Bolyai, Chem., 26 (1981) 52.
J. Zsakó, Gh. Marcu and M. Várhelyi, Rev. Roum. Chim., 27 (1982) 815.
J. Zsakó, E. Kékedy and Cs. Várhelyi, J. Thermal Anal., 1 (1969) 339.
T. Ozawa, Bull. Chem. Soc. Japan, 38 (1965) 1881.
J. Zsakó, Rev. Roum. Chim., 15 (1970) 693.
J. Zsakó, Cs. Várhelyi and E. Kékedy, Studia Univ. Babeş-Bolyai, Chem., 10 (1965) 7; Anal. Univ. Bucureşti, Chim., 14 (1965) 15; Proc. Anal. Chem. Conf., Budapest 1966, 353; J. Inorg. Nucl. Chem., 28 (1966) 2637; 32 (1970) 2999; Zh. Neorg. Khim., 13 (1968) 3279.
J. Zsakó, J. Phys. Chem., 72 (1968) 2406.
J. Zsakó, Cs. Várhelyi and G. Liptay, Proc. 4th ICTA, Budapest 1974, p. 825.
J. Zsakó, Cs. Várhelyi Jr., G. Marcu and G. Liptay, J. Therm. Anal. Cal., 55 (1999) 311.
J. Zsakó, E. Kékedy and Cs. Várhelyi, Rev. Roum. Chim., 15 (1970) 865.
J. Zsakó, J. Horák and Cs. Várhelyi, J. Thermal Anal., 20 (1981) 435.
J. Zsakó, J. Horák, Cs. Várhelyi and A. Benkő, Monatshn., 112 (1981) 945.
J. Zsakó, J. Thermal Anal., 15 (1979) 369.
J. Zsakó, J. Thermal Anal., 47 (1996) 1679.
J. Zsakó, J. Therm. Anal. Cal., 54 (1998) 921.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zsakó, J., Szilágyi, I., Simay, Á. et al. Kinetic analysis of TG data XXXVI: Influence of procedural variables upon the apparent kinetic parameters of the thermal deamination. Journal of Thermal Analysis and Calorimetry 69, 125–133 (2002). https://doi.org/10.1023/A:1019941824367
Issue Date:
DOI: https://doi.org/10.1023/A:1019941824367