Abstract
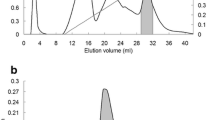
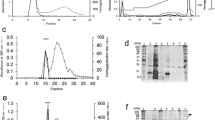
A lectin from Delonix regia (DRL) seeds was purified by gel filtration on Sephadex G-100 followed by ion-exchange chromatography on diethylaminoethyl-Sepharose and reverse-phase high-performance liquid chromatography on a C18 column. Hemagglutinating activity was monitored using rat erythrocytes. DRL showed no specificity for human erythrocytes of ABO blood groups. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) revealed a single protein in the presence of 0.1 M of dithiothreitol (DTT) and in nonreducing conditions. Native-PAGE showed that DRL is a monomer with a molecular mass of about 12 kDa, as determined by denaturing gel electrophoresis and gel filtration chromatography. An amino acid composition revealed the absence of cysteine residues, the presence of 1 mol methionine/mol protein and a high proportion of acidic amino acids and glycine. The N-terminal sequence of DRL was determined by Edman degradation, and up to 16 amino acid residues showed more than 90% homology with other lectins from the Leguminosae family. The optimal pH range for lectin activity was between pH 8.0 and 9.0, and the lectin was active up to 60°C. The lectin required Mn2+ for hemagglutinating activity and remained active after reduction with 0.1 M of DTT, but lost activity in the presence of 8 M of urea. Sodium metaperiodate had no effect on the activity of DRL.
Similar content being viewed by others
REFERENCES
Ahmad, S., Khan, R. H., and Ahmad, A. (1999). Biochim. Biophys. Acta 1427, 378-384.
Animashaun, T., Togun, R. A., and Hughes, R. C. (1994).Glycoconj. J. 11, 299-303.
Atta, A. M., Menezes, E. P., Pelxinho, S., and Souza-Atta, M. L. B. (1990). Braz. J. Biol. Res. 23, 191-194.
Baumann, C. M., Strosberg, A D., and Rüdiger, H. (1982). Int. Eur. J. Biochem. 122, 105-110.
Beuth, J., Ko, H. I., Pulverer, G., Uhlenbruck, G., and Pichlmaier, H. (1995). Glycoconj. J. 12, 1-6.
Bookbinder, L. H., Cheng, A., and Bleil, J. D. (1995). Science 269, 86-89.
Carrington, D. M., Auffret, A., and Hanke, D. E. (1985). Nature 313, 64-67.
Crispeels, M. J. and Raikhel, N. V. (1991). Plant Cell 3, 1-9.
Elgavish, S. and Shaanan, B. (1997). Trends Biochem. Sci. 22, 462-467.
Foriers, A., Lebrun, E., Van Rapenbusch, R., De Neve, R., and Strosberg, A. D. (1981). J. Biol. Chem. 256, 5550-5560.
Gatehouse, A M. R., Powell, K. S., Peumans, W. J., Van Damme, E. J. M., and Gatehouse, J. A. (1995). In: Lectins: Biochemistry Perspectives (Pusztai, A and Bardocz, S., eds), Taylor and Francis, London, pp. 35-57.
Goldstein, I. J. and Hayes, C. E. (1978). Adv. Carbohyd. Chem. Biochem. 37, 127-340.
Goldstein, I. J. and Poretz, R. D. (1986). In: The Lectins (Liener, I. E. and Sharon, N., eds), Academic Press, New York, pp. 33-247.
Henrikson, R. L. and Meredith, S. C. (1984). Anal. Biochem. 136, 65-71.
Hopp, T. P., Hemperly, J. J., and Cunningham, B. A. (1982). J. Biol. Chem. 257, 4473-4483.
Laemmli, U. K. (1970). Nature 227, 680-685.
Loris, R., Hamelryck, J., Bouckaert, J., and Wyns, L. (1998). Biochim. Biophys. Acta 1383, 9-36.
Lowry, O. H., Rosebrough, N. J., Fars, A. L., and Randall, R. J. (1951). J. Biol. Chem. 193, 165-275.
Mirkov, T. E., Wahlstrom, J. M., Hagiwara, K., Finardi-Filho, F., Kjemtrup, S., and Crisppels, M. J. (1994). Plant Mol. Biol. 26, 1103-1113.
Moreira, R. A., Castelo-Branco, C. C., Monteiro, A. C. O., Tavares, R. O., and Beltramini, L. M. (1998). Phytochemistry 47, 1183-1188.
Obregón-Henao, A., Gil, D. L., Gómez, D. I., Sanzón, F., Teale, J. M., and Restrepo, B. I. (2001). Mol. Biochem. Parasitol. 114, 209-215.
Ohsawa, F., Hirano, F., and Natori, S. (1990). J. Biochem. 107, 431-434.
Osborn, T. C., Brown, J. W. S., and Bliss, F. A. (1985). Theoret. Appl. Genet. 70, 22-31.
Ozeki, Y., Matsui, T., Nita, K., Kawauchi, H., Takayanagi, Y., and Titani, K. (1991). Biochem. Biophys. Res. Commun. 178, 407-413.
Perez, G. (1998). Int. J. Biochem. Cell Biol. 30, 843-853.
Peumans, W. L. and Van Damme, E. J. M. (1995). Plant Physiol. 109, 347-352.
Peumans, W. J. and Van Damme, E. J. M. (1998). Biotechnol. Genetic Eng. Rev. 15, 199-228.
Puy, D. J. Du., Phillipson, P. B., and Rabevohitra, R. (1995). Kew Bull. 50, 445-475.
Ray, S. and Chatterjee, B. P. (1995). Phytochemistry 40, 643-649.
Rini, J. M., Hardman, K. D., Einspahr, H., Suddath, F. L., and Carver, J. P. (1993). J. Biol. Chem. 268, 10126-10132.
Rini, J. M. (1995). Annu. Rev. Biophys. Biomol. Struct. 24, 551-577.
Rüdiger, H. (1997). In Glycosciences: Status and Perspectives (Gabius, H. J. and Gabius, S., eds.), Chapman & Hall, Weinhein, pp. 415-438.
Rüdiger, H. (1998). Acta Anat. 161, 130-152.
Sharon, N. and Lis, H. (1972). Science 177, 949-959.
Sharon, N. (1993). Trends Biol. Sci. 18, 221-226.
Sharon, N. and Lis, H. (1995). Essays Biochem. 30, 59-75.
Susselan, K. N., Bhatia, C. R., and Mitra, R. (1997). J. Biosc. 22, 439-455.
Suvachittanout, W. and Peutpaiboon, A. (1992). Phytochemistry 31, 4065-4070.
Teichberg, V. I., Aberdam, D., Ers, V., and Pinelli, E. (1988). J. Biol. Chem. 263, 14086-14092.
Utarabhand, P. and Akkayanont, P. (1995). Phytochemistry 38, 281-285.
Van Damme, J. M., Smeets, K., Torrekens, S., Van Leuven, F., and Peumans, W. J. (1993). Eur. J. Biochem. 217, 123-129.
Wang, G. F., Cao, Z. F., Zhou, A. M., and Zhao, Y. R. (2000). Int. J. Biochem. Cell Biol. 32, 873-878.
Wright, C. S. (1989). J. Mol. Biol. 209, 475-487.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pando, S.C., Macedo, M.L.R., Freire, M.G.M. et al. Biochemical Characterization of a Lectin from Delonix regia Seeds. J Protein Chem 21, 279–285 (2002). https://doi.org/10.1023/A:1019797320348
Published:
Issue Date:
DOI: https://doi.org/10.1023/A:1019797320348