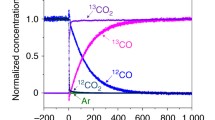
Abstract
The adsorption and reaction of CO, CO2 and O2 on TiO2 and Au/TiO2 have been studied using a mass spectrometric method which can detect processes occurring on a time scale of seconds. Adsorption of CO on TiO2 at 300 K is rapidly reversible and less on reduced samples than oxidised ones indicating that the adsorption sites are oxide ions. The amount adsorbed reversibly on reduced Au/TiO2 is less still, consistent with enhanced reduction, but additional amounts adsorb irreversibly at a slower rate. The amount of CO2 adsorbed under similar conditions is also greater on TiO2 than reduced Au/TiO2 and approximately one order of magnitude greater than that of CO. However, adsorption of O2 is undetectable on the time scale of the measurement. Exposure of Au/TiO2 to mixtures of CO and O2 results in near instantaneous generation of CO2 although its appearance is attenuated by adsorption. Adsorption of CO occurs concurrently in a way similar to that seen with CO alone except that the amount of the more slowly adsorbed form seems less. This suggests that it is the form utilised in catalysis. Oxygen uptake beyond that generating CO2 is appreciable during the initial stages of exposure to reaction mixtures and this capacity is enhanced if one or other reactant is removed and then reintroduced, possibly due to the generation of reducible interface sites. It is concluded that the remarkable activity of Au/TiO2 for CO oxidation at ambient temperature resides in a very high turnover frequency on sites at the interface between the metal and oxide.
Similar content being viewed by others
References
R.K. Herz, NASA Conference Publication 3076 (1990) 21.
S.D. Gardner, G.B. Hoflund, D.R. Schryer, J. Schryer, B.T. Upchurch and E.J. Kielin, Langmuir 7 (1991) 2135.
J.H. Kolts, S.H. Brown and P.A. Tooley, US Patent4,943,550(1990).
T. Kobayashi, M. Haruta, H. Sano and M. Nakane, Sensors and Actuators 13 (1988) 339.
M. Haruta, S. Tsubota, T. Kobayashi, H. Kageyama, M.J. Genet and B. Delmon, J. Catal. 144 (1993) 175.
S. Tsubota, D. Cunningham, Y. Bando and M. Haruta, Stud. Surf. Sci. Catal. 77 (1993) 325.
S.D. Lin, M. Bollinger and M.A. Vannice, Catal. Lett. 17 (1993) 245.
M.A. Bollinger and M.A. Vannice, Appl. Catal. B 8 (1996) 417.
S.J. Tauster, ACS Symposium Series 298 (1986) 1.
S.J. Tauster, S.C. Fung, R.T.K. Baker and J.A. Horsley, Science 211 (1981) 1121.
G.S. Lane and E.E. Wolf, J. Catal. 105 (1987) 386.
K. Tanaka and J.M. White, J. Phys. Chem. 86 (1982) 3977.
A.G. Shastri, A.K. Datye and J. Schwank,J. Catal. 78 (1984) 265.
S.D. Lin and M.A. Vannice, Catal. Lett. 10 (1991) 1.
S.D. Gardner, G.B. Hoflund, M.R. Davidson, H.A. Laitenen, D.R. Schryer and B.T. Upchurch, Langmuir 7 (1991) 2140.
J.S. Smith, P.A. Thrower and M.A. Vannice, J. Catal. 68 (1981) 270.
J.R. Anderson and K.C. Pratt, Introduction to Characterisation and Testing of Catalysts (Academic Press, North Ryde, 1985).
N.J. Ossipoff and N.W. Cant, Catal. Today 36 (1997) 125.
C. Morterra, J. Chem. Soc., Faraday Trans. I 84 (1988) 1617.
P. Dumas, R.G. Tobin and P.L. Richards, Surf. Sci. 171 (1986) 579.
J. France and P. Hollins, J. Electr. Spectros. Rel. Phenom. 64/65 (1993) 251.
F. Boccuzzi, A. Chiorino, S. Tsubota and M. Haruta, J. Phys. Chem. 100 (1996) 3625.
F. Boccuzzi, A. Chiorino, S. Tsubota and M. Haruta, Catal. Lett. 29 (1994) 225.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ossipoff, N.J., Cant, N.W. Adsorption and reactivity during low temperature oxidation of carbon monoxide on Au/TiO2 catalysts studied by rapid response mass spectrometry. Topics in Catalysis 8, 161–169 (1999). https://doi.org/10.1023/A:1019161111188
Issue Date:
DOI: https://doi.org/10.1023/A:1019161111188