Abstract
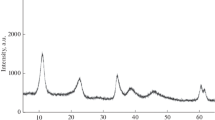
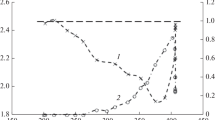
A study of the hydrogenation of solid CaCO3 has shown that the addition of Pd and Ir catalysts causes a change in the gaseous product and, consequently, in the kinetics of the reaction. In the absence of catalyst, CO is formed at higher than 700 K with the activation energy of 236 kJ/mol and a nearly half order with respect to H2 pressure, which is explained by the mechanism consisting of predecomposition and reduction. In the presence of the catalysts, CH4 is exclusively formed even at 573 K at which the equilibrium decomposition pressure of CaCO3 is extremely low, 1.1 × 10−5 Torr. Activation energies found in the range 105–118 kJ/mol and the H2-pressure dependence of the initial rate suggest the direct interaction of CaCO3 with spilt-over hydrogen atoms.
Similar content being viewed by others
References
A. Tsuneto, A. Kudo, N. Saito and T. Sakata, Chem. Lett. (1992) 831.
J.E. Benson and M. Boudart, J. Catal. 4 (1965) 704.
J.E. Benson, H.S. Hwang and M. Boudart, J. Catal. 30 (1973) 146.
A.W.D. Hills, Chem. Eng. Sci. 23 (1968) 297.
W.C. Conner, Jr., G.M. Pajonk and S.J. Teichner, in: Advances in Catalysis, Vol. 34, eds. D.D. Eley, H. Pines and P.B. Weisz (Academic Press, New York, 1986).
K.J. Hill and E.R.S. Winter, J. Phys. Chem. 60 (1956) 1361.
D. Beruto and A.W. Searcy, J. Chem. Soc. Faraday Trans. I 70 (1974) 2145; P.K. Gallagher and D.W. Johnson, Jr., Thermochim. Acta 6 (1973) 67.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yoshida, N., Hattori, T., Komai, E. et al. Methane formation by metal-catalyzed hydrogenation of solid calcium carbonate. Catalysis Letters 58, 119–122 (1999). https://doi.org/10.1023/A:1019017615013
Issue Date:
DOI: https://doi.org/10.1023/A:1019017615013