Abstract
Purpose. Establishment of the pharmacokinetic profile of methotrexate (MTX) in the extracellular fluid (ECF) of a brain C6-glioma in rats.
Methods. Serial collection of plasma samples and ECF dialysates after i.v. infusion of MTX (50 or 100 mg/kg) for 4 h. HPLC assay.
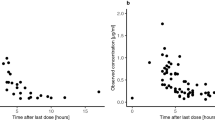
Results. Histological studies revealed the presence of inflammation, edema, necrosis, and hemorrhage in most animals. In vivo recovery (reverse dialysis) was 10.8 ± 5.3%. MTX concentrations in tumor ECF represented about 1−2% of the plasma concentrations. Rapid equilibration between MTX levels in brain tumor ECF and plasma. ECF concentrations almost reached steady-state by the end of the infusion (4 h), then decayed in parallel with those in plasma. Doubling of the dose did not modify MTX pharmacokinetic parameters (t1/2α, t1/2β, MRT, fb, Vd, and CLT), except for a 1.7-fold increase of AUCPlasma and a 3.8-fold increase in AUCECF which resulted in a 2.3-fold increase in penetration (AUCECF/AUCPlasma). In spite of an important interindividual variability, a relationship between MTX concentrations in plasma and tumor ECF could be established from mean pharmacokinetic parameters.
Conclusions. High plasma concentrations promote the penetration of MTX into brain tissue. However, free MTX concentrations in tumor ECF remain difficult to predict consistently.
Similar content being viewed by others
REFERENCES
E. Chatelut, H. Roche, Y. Plusquellec, F. Peyrille, J. DeBiasi, A. Pujol, P. Canal, and G. Houin. Pharmacokinetic modeling of plasma and cerebrospinal fluid methotrexate after high-dose intravenous infusion in children. J. Pharm. Sci. 80: 730-734 (1991).
D. Nierenberg, R. Harbaugh, L. Herbert Mauer, T. Reeder, G. Scott, J. Fratkin, and E. Newman. Continuous intratumoral infusion of methotrexate for recurrent glioblastoma: a pilot study. Neurosurg. 28:752-761 (1991).
L. Slordal, R. Jaeger, J. Kjaeve, and J. Aarbakke. Pharmacokinetics of 7-hydroxy-methotrexate and methotrexate in the rat. Pharmacol. Toxicol. 63:81-84 (1988).
R. K. Jain, J. Wei, and P. M. Gullino. Pharmacokinetics of methotrexate in solid tumors. J. Pharmacoki. Biopharm. 7:181-194 (1979).
P. A. Miglioli, V. Businaro, F. Manoni, and T. Berti. Tissue distribution of methotrexate in rats. Comparison between intravenous injection as bolus or drip infusion. Drugs Exptl. Clin. Res. 11:175-279 (1985).
E. C. M. de Lange, J. D. de Vries, C. Zurcher, M. Danhof, A. G. de Boer, and D. D. Breimer. The use of intracerebral microdialysis for the determination of pharmacokinetic profiles of anticancer drugs in tumor-bearing rat brain. Pharm. Res. 12:1924-1931 (1995).
D. Devineni, A. Klein-Szanto, and J. M. Gallo. In vivo microdialysis to characterize drug transport in brain tumors: analysis of methotrexate uptake in rat glioma-2 (RG-2)-bearing rats. Cancer Chemother. Pharmacol. 38:499-507 (1996).
P. Benda, J. Lightbody, G. Sato, L. Levine, and W. Sweet. Differentiated rat glial cell strain in tissue culture. Science 161:370-371 (1968).
S. I. Rapoport, W. R. Fredericks, K. Ohno, and K. D. Pettigrew. Quantitative aspectfs of reversible osmotic opening of the blood-brain barrier. Am. J. Physiol. 238:421-431 (1980).
A. Le Quellec, S. Dupin, P. Genissel, S. Saivin, B. Marchand, and G. Houin. Microdialysis probes calibration: Gradient and tissue dependent changes in No Net Flux and Reverse Dialysis methods. J. Pharmacol. Toxicol. Meth. 33:11-16 (1995).
M. Gibaldi and D. Perrier. Pharmacokinetics, Marcel Dekker, New York, 1982.
D. R. Groothuis, J. M. Fischer, J. F. Pasternak, R. G. Blasberg, N. A. Vick, and D. D. Bigner. Reagional measurements of the blood-to-tissue transport in experimental RG-2 rat gliomas. Cancer Res. 43:3368-3373 (1983).
P. A. Stewart, K. Hayakawa, E. Hayakawa, C. L. Farrell, and R. F. Del Maestro. A quantitative study of blood-brain barrier permeability ultrastructure in anew rat glioma model. Acta. Neuropathol. (Berl). 67:96-102 (1985).
J. J. Bernstein, W. J. Goldberg, E. R. Laws, D. Conger, V. Morreale, and L. R. Wood. C6 glioma cell invasion and migration of rat brain after neural homografting: ultrastructure. Neurosurg. 26:622-628 (1990).
V. M. Morreale, B. H. Herman, V. Der-Minassian, M. Palkovits, P. Klubes, D. Perry, A. Csiffary, and A. P. Lee. A brain-tumor model utilizing stereotactic implantation of a permanent cannula. J. Neurosurg. 78:959-965 (1993).
N. Kobayashi, N. Allen, N. R. Clendenon, and L. W. Ko. An improved rat brain-tumor model. J. Neurosurg. 53:808-815 (1980).
K. Yamada, T. Hayakawa, Y. Ushio, N. Arita, A. Kato, and H. Mogami. Regional blood flow and capillary permeability in the ethylnitrosourea-induced rat glioma. J. Neurosurg. 55:922-928 (1981).
B. R. Deane and P. L. Lantos. The vasculature of experimental brain tumors. Part 2. A quantitative assessment of morphological abnormalities. J. Neurol. Sci. 49:67-77 (1981).
N. A. Vick, J. D. Khandekar, and D. D. Bigner. Chemotherapy of brain tumors: the 'blood-brain barrier' is not a factor. Arch. Neurol. 34:523-526 (1977).
R. M. Bremnes, L. Slordal, E. Wist, and J. Aarbakkle. Dose-dependent pharmacokinetics of methotrexate and 7-hydroxymethotrexate in the rat in vivo. Cancer Res. 49:6359-6364 (1989).
P. O. Ekstrom, A. Andersen, D. J. Warren, K. E. Giercksky, and L. Slordal. Pharmacokinetics of different doses of methotrexate at steady state by microdialysis in a rat model. Cancer Chemother. Pharmacol. 36:283-289 (1995).
E. Scheufler. Evidence for nonlinear pharmacokinetics of methotrexate in the rat. Pharmacol. 25:51-56 (1982).
M. Hammarlund-Udenaes, L. K. Paalzow, and E. C. M. de Lange. Drug equilibration across the blood-brain barrier: Pharmacokinetic considerations based on the microdialysis method. Pharm. Res. 14:128-134 (1997).
W. R. Shapiro, R. M. Voorhies, E. M. Hiesiger, P. B. Sher, G. A. Basler, and L. E. Lipschutz. Pharmacokinetics of tumor cell exposure to 14C methotrexate after intracarotid administration without and with hyperosmotic opening of the blood-brain and blood-tumor barriers in rat brain tumors: a quantitative autoradiographic study. Cancer Res. 48:694-701 (1988).
D. D. Shen and D. L. Azarnoff. Clinical pharmacokinetics of methotrexate. Clin. Pharmacokinet. 3:1-13 (1978).
W. Bleyer and D. G. Pollack. Clinical studies on the central nervous system pharmacology of methotrexate. In: H. M. Pinedo (ed.) Clinical pharmacology of antineoplastic drugs. North Holland Biomedical Press, Amsterdam, 1978, pp. 115-130.
W. E. Evans, W. R. Crom, and J. C. Yalowich. Methotrexate. In W. E. Evans J. J. Schentag, and W. J. Jusko (eds.), Applied pharmacokinetics: Principles of therapeutic drug monitoring, 4th ed., Applied therapeutics, Washington, 1986, pp. 1009-1056.
J. H. Schornagel and J. G. McVie. The clinical pharmacology of methotrexate. Cancer Treat. Rev. 10:53-75 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dukic, S., Heurtaux, T., Kaltenbach, M.L. et al. Pharmacokinetics of Methotrexate in the Extracellular Fluid of Brain C6-Glioma After Intravenous Infusion in Rats. Pharm Res 16, 1219–1225 (1999). https://doi.org/10.1023/A:1018945529611
Issue Date:
DOI: https://doi.org/10.1023/A:1018945529611