Abstract
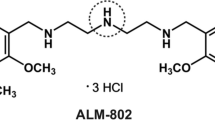
The pharmacokinetics of a novel antiarrhythmic drug, actisomide, were examined in the rat, dog, monkey, and human. The terminal half-life of actisomide was similar (1.15–1.89 hr) across species, regardless of dose. The total plasma clearance was higher in the monkey (13.5–16.4 mL/min/kg) than in the dog (9.01–9.32 mL/min/kg), rat (8.6–9.8 mL/min/kg), or human (6.79 ± 1.07 mL/min/kg). Excretion of the parent drug was higher in urine than in feces in the dog and rat, whereas in the monkey and human, urinary and fecal excretions of actisomide were similar. In humans, atypical plasma concentration–time curves with double peak concentrations were observed following oral doses. Systemic availability of actisomide was higher in the dog than in the rat, monkey, and human. Further, the systemic availability appeared to increase with dose in the rat and monkey. The species-dependent systemic availability appeared to be due primarily to species-dependent absorption of actisomide, and not to species-dependent first-pass metabolism, biliary excretion, and/or renal elimination. The absorption of actisomide in the rat and its in vitro uptake in CaCo-2 cells were pH dependent. The higher systemic availability of actisomide observed in the dog may be due partly to the higher pH in the gastrointestinal (GI) tract of the dog. However, the pH differences in the GI tract of the different species alone did not appear to be enough to explain the difference in systemic availability of actisomide.
Similar content being viewed by others
REFERENCES
S. M. Garthwaite, F. R. Hartly, L. G. Frederick, and C. S. Cook. Efficacy and plasma concentrations of SC-36602 in canine models of ventricular arrhythmia. J. Cardiovasc. Pharmacol. 13:218–226 (1989).
S. Garthwaite, C. Cook, D. Semler, and R. Dean. Actisomide. Cardiovasc. Drug Rev. 7:52–67 (1989).
P. J. Sinko and G. L. Amidon. Characterization of the oral absorption of beta-lactam antibiotics, cephalosporins: Determination of intrinsic membrane absorption parameters in the rat intestine in situ. Pharm. Res. 5:645–650 (1988).
Z. Ruben, D. C. Dodd, K. J. Rorig, and S. N. Anderson. Disobutamide: A model agent for investigating intracellular drug storage. Toxicol. Appl. Pharmacol. 97:57–71 (1989).
C. M. Metzler, G. K. Elfring, and A. J. McEween. A package of computer programs for pharmacokinetic modeling. Biometrics 30:562–563 (1974).
B. I. Hirschowitz. Apparent kinetics of histamine dose-responsive gastric water and electrolyte secretion in the dog. Gastroenterology 54:514–522 (1968).
H. Petersen and M. I. Grossman. Stimulation of gastric acid secretion of dinaprit in unanesthetized rats. Agents Actions 8:566–567 (1978).
H. W. Smith. Observations on the flora of the alimentary tract of animals and factors affecting its composition. J. Pathol. Bacereol. 89:95–122 (1965).
N. I. McNeil and K. L. E. Ling. Large intestinal mucosal surface pH in rat and man. In E. Skadhauge and K. Herztze (eds.), Intestinal Absorption and Secretion, MTP Press, Boston, 1984, pp. 103–109.
M. Gibaldi. Biopharmaceutics and Clinical Pharmacokinetics, Lea & Febieger, Philadelphia, 1977.
K. J. Rorig, Z. R. Ruben, and S. N. Anderson. Structural determinants of cationic amphiphilic amines which induce clear cytoplasmic vacuoles in culture cells (42462). Proc. Soc. Exp. Biol. Med. 184:165–171 (1987).
B. N. Besai, C. S. Cook, W. D. Claypool, and S. M. Garthwaite. Bidisomide. Drugs Future 17:374–376 (1992).
C. G. Regardh, P. Lundborg, M. Gabrielesson, A. Heggelund, and K. Kylberg-Hassen. Pharmacokinetics of a single intravenous and oral dose of pafenolol—A beta-adrenoceptor antagonist with atypical absorption and disposition properties—in man. Pharm. Res. 7:1222–1227 (1990).
R. F. Bergstrom, D. R. Kay, T. M. Harkom, and J. G. Wagner. Penicillamine kinetics in normal subjects. Clin. Pharmacol. Ther. 30:404–413 (1981).
T. Funaka, S. Furuta, and N. Kaneniwa. Discontinuous absorption of cimetidine. Int. J. Pharm. 31:119–123 (1986).
C. K. Shim and J. S. Hong. Inter-and intrasubject variations of ranitidine pharmacokinetics after oral administration to normal subjects. J. Pharm. Sci. 71:990–994 (1989).
Y. Plusquellec, G. Campistron, S. Staveris, J. Barre, L. Jung, J. P. Tillement, and G. Houin. A double-peak phenomenon in pharmacokinetics of veralipride after oral administration: A double-site model for drug absorption. J. Pharmacokin. Biopharm. 15:225–239 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cook, C.S., Rozek, L.F., Stolzenbach, J. et al. Pharmacokinetics of a Novel Antiarrhythmic Drug, Actisomide. Pharm Res 10, 427–433 (1993). https://doi.org/10.1023/A:1018900725050
Issue Date:
DOI: https://doi.org/10.1023/A:1018900725050