Abstract
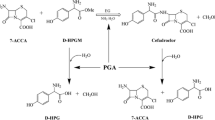
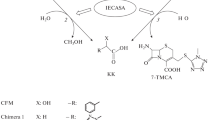
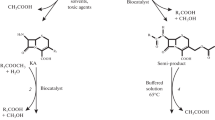
Conversion of cephalosporin C into glutaryl 7-aminocephalosporanic acid was catalysed by D-aminoacid oxidase from Trigonopsis variabilis, covalently immobilized on the polystyrenic resin Duolite A365. Spontaneous degradation of substrates was limited without depressing enzymatic activity at the optimum reaction pH 8.0. The highest product yield was 1.77 mmol per g of biocatalyst, attained at 15¡C in both batch stirred and fluidized-bed reactors.
Similar content being viewed by others
References
Alfani F., Cutarella N., Gallifuoco A., Cantarella M., Bianchi D. and Bosetti A. (1993). VI European Congress on Biotechnology, Firenze (Italy) 13–17 June 4, TH057.
Alfani F., Cutarella N., Gallifuoco A., Cantarella M., Golini P., Franzosi G. and Bianchi D. (1995). Annals N.Y. Acad. of Sciences 750, 491-495.
Butò S., Pollegioni L., D'Angiuro L. and Pilone M.S. (1994). Biotech. Bioeng 44, 1288-1294.
Golini P., Bianchi D., Battistel E., Cesti P. and Tassinari R. (1995). Enzyme Microb. Technol. 17, 324-329.
Nikolov A. and Danielsson B. (1994). Enzyme Microb. Technol. 16, 1037-1040.
Vicenzi J.T. and Hansen G.T. (1993). Enzyme Microb. Technol. 15, 281-285.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Alfani, F., Cantarella, M., Cutarella, N. et al. Enzymatic conversion of cephalosporin C into glutaryl 7-aminocephalosporanic acid. A study in different reactor configurations. Biotechnology Letters 19, 175–178 (1997). https://doi.org/10.1023/A:1018324700707
Issue Date:
DOI: https://doi.org/10.1023/A:1018324700707