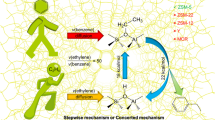
Abstract
The ring-opening reactions of methyloxirane and ethyloxirane on ZSM-5 catalysts are dramatically different in spite of otherwise identical reaction conditions. Even though both reactants undergo single ring opening providing isomers of the starting compound, the major transformation route of methyloxirane is dimerization resulting in dioxolane and dioxane derivatives of various kinds. This reaction pathway, however, is completely lacking in the case of ethyloxirane. Molecular modeling revealed that possible dimers of ethyloxirane are too bulky to be formed and diffuse through the channel system of the ZSM-5 structure.
Similar content being viewed by others
REFERENCES
S.E. Denmark, P.A. Barsanti, K.-T. Wong, R.A. Stavenger: J. Catal., 63, 2428 (1998).
M. Bartók: in The Chemistry of Functional Groups Supplement E2: The Chemistry of Hydroxyl, Ether and Peroxide Groups: (S. Patai, Ed.), Chap. 15, p. 843. Wiley, Chichester, Brisbane, New York, Toronto, Singapore 1993.
A. Fási, I. Pálinkó: J. Catal., 181, 28 (1999).
H.G. Karge, H.K. Beyer: Stud. Surf. Sci. Catal., 69, 43 (1991).
Á. Molnár, I. Bucsi, M. Bartók, G. Resofszki, Gy. Gáti: J. Catal., 129, 303 (1991).
Ch. Baerlocher, W.M. Meier, D.H. Olson: in Atlas of Zeolite Framework Types, p. 185. Elsevier, Amsterdam, London, New York, Oxford, Paris, Shannon, Tokyo 2001.
M.J.S. Dewar, E.G. Zoebisch, E.F. Healy, J.J.P. Stewart: J. Am. Chem. Soc., 107, 3902 (1985).
Hyperchem 6.02, Hypercube, Inc., 1999, Gainesville, FL, USA.
A. Bondi: J. Phys. Chem., 68, 441 (1964).
A. Fási, I. Pálinkó, I. Kiricsi: J. Catal., 188, 385 (1999).
A. Fási, Á. Gömöry, I. Pálinkó, I. Kiricsi: J. Catal., 200, 340 (2001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fási, A., Pálinkó, I. & Kiricsi, I. Effects of Substituents on the Ring-Opening Pathways of Oxiranes on ZSM-5 Zeolites. Reaction Kinetics and Catalysis Letters 74, 187–194 (2001). https://doi.org/10.1023/A:1017960321702
Issue Date:
DOI: https://doi.org/10.1023/A:1017960321702