Abstract
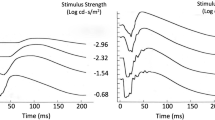
Accurate interpretation of electroretinograms (ERGs) requires knowledge of effects of axial myopia on ERG responses. Our purpose was to derive expected changes of ERG responses according to axial length, to stimulus conditions that conform to the International Society for Clinical Electrophysiology of Vision (ISCEV) Standard for Electroretinography. ERGs from 60 subjects were recorded. The subjects were assigned to one of three groups according to the level of myopia. Thirty-three subjects had high myopia (−6.00 D to −14.50 D; mean age, 31 years), eight had mild myopia (−3.00 D to −5.00; mean age, 28 years), and 19 had a small refractive error (+0.75 D to −2.75 D; mean age, 27 years). No subjects had myopic retinopathy. Stimulus-response curves were fitted to dark-adapted b-wave amplitudes and maximum amplitude and semi-saturation constants derived. Axial lengths, measured with A scan ultrasound, ranged from 22.2 mm to 30.0 mm. Analysis of variance and post hoc t-tests revealed significant difference between subjects with high myopia and subjects with small refractive error for ERG amplitude data. There were no significant differences between the three groups for implicit times, the ratio of b- to a-wave and semi-saturation constant. There is linear reduction in the logarithmic transform of ERG amplitude with increasing axial length, related more to axial length than refractive error. We provide relative slope and intercept values, allowing labs to derive expected ERG amplitudes according to axial length. These derivations are valid for persons with no retinopathy.
Similar content being viewed by others
References
Birch DC, Anderson JL. Standardized full-field electroretinography. Normal values and their variations with age. Arch Ophthalmol 1992; 110: 1571–6.
Westall CA, Panton CM, Levin AV. Time courses for maturation of electroretinogram responses from infancy to adulthood. Doc Ophthalmol 1999; 96: 355–79.
Weleber RG. The effect of age on human cone and rod ganzfeld electroretinograms. Invest Ophthalmol Vis Sci 1981; 20(3): 392–9.
Fulton AB, Hansen RM. Photoreceptor function in infants and children with a history of mild retinopathy of prematurity. J Opt Soc Am A 1996; 13(3): 566–71.
Niemeyer G. Pharmacological effects in retinal electrophysiology. In: Heckenlively JR, Arden GB, eds. Principles and Practice of Clinical Electrophysiology of Vision. St. Louis, MO: Mosby-Yearbook Inc., 1991: 151–62.
Sannita WG. Neuropsychiatric drug effects on the visual nervous system. In: Heckenlively JR, Arden GB, eds. Principles and Practice of Clinical Electrophysiology of Vision. St. Louis, MO: Mosby-Yearbook Inc., 1991: 167–73.
Fishman GA. Retinal toxicity with the use of chloroquine or hydroxychloroquine. In: Heckenlively JR, Arden GB, eds. Principles and Practice of Clinical Electrophysiology of Vision. St. Louis, MO: Mosby-Yearbook Inc., 1991: 594–9.
Marmor MF. Retinal toxicity from thioridazine and other phenothiazines. In: Heckenlively JR, Arden GB, eds. Principles and Practice of Clinical Electrophysiology of Vision. St. Louis, MO: Mosby-Yearbook Inc., 1991: 600–6.
Pallin E. The influence of the axial size of the eye on the size of the recorded b-potential in the clinical single-flash electroretinogram. Acta Ophthalmol 1969; 191 (suppl): 1–57.
Blach R, Jay BKH. Electrical activity of the eye in high myopia. Br J Ophthalmol 1966; 50(11): 629–41.
Perlman I, Meyer E, Haim T, Zonis S. Retinal function in high refractive error assessed electroretinographically. Br J Ophthalmol 1984; 68(2): 79–84.
Perlman I: Relationship between the amplitudes of the b wave and the a wave as a useful index for evaluating the electroretinogram. Br J Ophthalmol 1983; 67(7): 443–8.
Chen JF, Elsner AE, Burns SA, Hansen RM, Lou PL, Kwong KK, et al. The effect of eye shape on retinal responses. Clin Vision Sci 1992; 7(6): 520–30.
Kawabata H, Adachi-Usami E. Multifocal electroretinogram in myopia. Invest Ophthalmol Vis Sci 1997; 38(13): 2844–51.
Yamamoto S, Nitta K, Kamiyama M. Cone electroretinogram to chromatic stimuli in myopic eyes. Vision Res 1997; 37(15): 2157–9.
Aladjov S, Denev G, Penov G. The influence of galvanic current on the electroretinogram of the myopic eye. In: The clinical value of electroretinography, ISCERG Symp.; 1966, 1968. Ghent: Karger, Basel/New York, 1966: 444–50.
Prijot E, Colmant I, Marechal-Courtois C. Electroretinography and Myopia. In: Francois J, ed. Fifth Symposium of the International Society for Clinical Electroretinography (ISGERG); 1966, 1968. Ghent, Belgium: Karger, New York, 1966: 440–3.
Anonymous. Standard for clinical electroretinography. International Standardization Committee. Arch Ophthalmol 1989; 107: 816–9.
McGraw PV, Winn B. Glasgow Acuity Cards: a new test for the measurement of letter acuity in children. Ophthalm Physiol Opt 1993; 13: 400–4.
Mollon JD, Astell S, Reffin JP. A minimalist test of colour vision. In: Drum B, Moreland, JD, Serra A, eds. Documenta Ophthalmologica Proceedings Series. Colour vision deficiencies X. Dordrecht: Kluwer Academic Publ 1991: 59–67.
Marmor MF, Zrenner E. Standard for clinical electroretinography (1994 update). Doc Ophthalmol 1995; 89: 199–210.
Severns ML, Johnson MA. The care and fitting of Naka-Rushton functions to electroretinographic intensity-response data. Doc Ophthalmol 1993; 85: 135–50.
Peachey NS, Fishman GA, Derlacki DJ, Alexander KR. Rod and cone dysfunction in carriers of X-linked retinitis pigmentosa. Ophthalmology 1988; 95(5): 677–85.
Jacobi PC, Miliczek KD, Zrenner E. Experiences with the international standard for clinical electroretinography: normative values for clinical practice, interindividual and intraindividual variations and possible extensions [see comments]. Doc Ophthalmol 1993; 85(2): 95–114.
Fledelius HC. Ophthalmic changes from age of 10 to 18 years. A longitudinal study of sequels to low birth weight. IV. Ultrasound oculometry of vitreous and axial length. Acta Ophthalmol (Copenh) 1982; 60(3): 403–11.
Koretz JF, Rogot A, Kaufman PL. Physiological strategies for emmetropia. Trans Am Ophthalmol Soc 1995; 93: 105–18.
Murayama K, Sieving PA. Different rates of growth of monkey and human photopic a-, b-, and d-waves suggest two sites of ERG light adaptation. Clin Vision Sci 1992; 7(5): 385–92.
Huang B, Karwoski C. Light-evoked increase in subretinal space (SRS) in frog retina. Invest Ophthalmol Vis Sci 1990; 31(Suppl): 71.
Fulton AB, Hansen RM. Electroretinogram responses and refractive errors in patients with a history of retinopathy of prematurity. Doc Ophthalmol 1995; 91(2): 87–100.
Wachtmeister L. Oscillatory potentials in the retina: what do they reveal. Prog Retin Eye Res 1998; 17(4): 485–521.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Westall, C.A., Dhaliwal, H.S., Panton, C.M. et al. Values of electroretinogram responses according to axial length. Doc Ophthalmol 102, 115–130 (2001). https://doi.org/10.1023/A:1017535207481
Issue Date:
DOI: https://doi.org/10.1023/A:1017535207481