Abstract
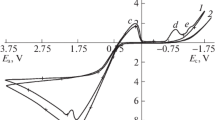
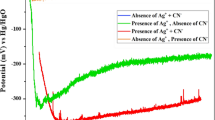
Data on the influence the solution's ionic composition and amino acids (histidine, methionine) have on the anodic dissolution of silver are presented. The dissolution is most effective in intensively agitated nitrate solutions containing methionine in excess of 0.05 M (current efficiency reaches 75%). By analogy with the gold dissolution, acceleration of the silver dissolution is attributed to oxidized methionine complexing with metals.
Similar content being viewed by others
REFERENCES
Tarasevich, E.M., Bogdanovskaya, V.A., and Yarlykov, M.M., Elektrokhimiya, 1984, vol. 20, p. 1093.
Safronov, A.Yu., Tarasevich, M.R., Bogdanovskaya, V.A., and Chernyak, A.S., Elektrokhimiya, 1983, vol. 19, p. 423.
Safronov, A.Yu., Tarasevich, M.R., Bogdanovskaya, V.A., and Chernyak, A.S., Elektrokhimiya, 1983, vol. 19, p. 1185.
Tumanova, E.A. and Safronov, A.Yu., Elektrokhimiya, 1998, vol. 34, p. 170.
Spravochnik khimika (A Chemist's Handbook), Leningrad: Khimiya, 1952, vol. 3.
Inorganic Biochemistry, Eichhorn, G.H., Ed., Amsterdam: Elsevier, 1973.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tarasevich, M.R., Chernysheva, L.S., Bogdanovskaya, V.A. et al. Electrooxidation of Silver in the Presence of Some Amino Acids. Russian Journal of Electrochemistry 37, 440–442 (2001). https://doi.org/10.1023/A:1016650712046
Issue Date:
DOI: https://doi.org/10.1023/A:1016650712046