Abstract
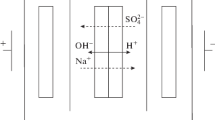
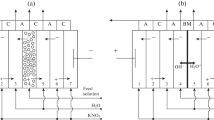
Bipolar membranes intended for the generation of hydrogen and hydroxyl ions in order to convert bipolar ions of amino acid into cations or anions migrating through cation- or anion-exchange membranes under a gradient of electric potential applied to the electromembrane system are studied. The transport processes occurring in glycine and sucrose solutions under electrodialysis with bipolar and unipolar ion-exchange membranes are examined. The proposed method permits the separation of a mixture of amino acids and sugars.
Similar content being viewed by others
REFERENCES
Shaposhnik, V.A., Eliseeva, T.V., and Selemenev, V.F., Elektrokhimiya, 1993, vol. 29, p. 794.
Shaposhnik, V.A., Selemenev, V.F., Terent'eva, N.P., and Oros, G.Yu., Zh. Prikl. Khim. (Leningrad), 1988, vol. 61, p. 1183.
Shaposhnik, V.A. and Eliseeva, T.V., J. Membr. Sci., 1999, vol. 161, p. 223.
Ionitovye membrany. Granulyaty. Poroshki: Katalog (The Ionite Membranes, Granulates, and Powders: A Catalogue), Pashkov, A.B., Ed., Moscow: NIITEKhim, 1977, p. 31.
Albert, A. and Serjeant, E., Ionization Constants of Acids and Bases, New York: Wiley, 1962.
Terminology for Membrane and Membrane Processes, J. Membr. Sci., 1996, vol. 120, p. 150.
Simons, R., Electrochim. Acta, 1984, vol. 29, p. 151.
Eliseeva, T.V. and Shaposhnik, V.A., Elektrokhimiya, 2000, vol. 36, p. 73.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eliseeva, T.V., Tekuchev, A.Y., Shaposhnik, V.A. et al. Electrodialysis of Amino Acid Solutions with Bipolar Ion-Exchange Membranes. Russian Journal of Electrochemistry 37, 423–426 (2001). https://doi.org/10.1023/A:1016642510229
Issue Date:
DOI: https://doi.org/10.1023/A:1016642510229