Abstract
Purpose. The purpose of this study was to investigate the utility of a purified, semisynthetic saponin, DS-1, prepared by deacylation of a naturally occurring saponin from the bark of the Quillaja saponaria Molina tree, as a permeation enhancer for mucosal delivery of the aminoglycosides, gentamicin and tobramycin.
Methods. Gentamicin or tobramycin formulations, with and without DS-1, were administered to rats nasally, ocularly, and rectally. Serum aminoglycoside levels following mucosal application were compared with those administered intramuscularly. Gentamicin formulations, with and without DS-1, were administered intranasally to mice 60 minutes after a lethal bacterial challenge. To ascertain nasal irritation potential, DS-1 nosedrops were administered to rats twice daily for 7 days in the right nostril only. Comparison of the left (internal control) and right nostril was made with a control group that received only buffer.
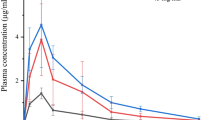
Results. Significant transport across mucous membranes was only observed in formulations containing DS-1. This effect on drug delivery was transient. Administration of an intranasal gentamicin/ DS-1 formulation reversed the lethal bacterial challenge in mice, demonstrating that biological activity was retained after absorption. Nasal irritation was not observed in groups receiving DS-1 nose-drops, which were identical to control groups.
Conclusions. DS-1 has potential as a transmucosal delivery agent for the aminoglycoside antibiotics.
Similar content being viewed by others
REFERENCES
A. G. Gilman, T. W. Rall, A. S. Nies, and P. Taylor. Goodman and Gilman's The Pharmacological Basis of Therapeutics, 8th edition, Pergamon Press, New York, 1990.
D. M. Poretz. Outpatient Use of Intravenous Antibiotics. Amer. J. Med. 97 (suppl. 2A):1–2 (1994).
R. J. Roberts. Issues and Problems Associated with Drug Delivery in Pediatric Patients. J. Clin. Pharmacol. 34:723–724 (1994).
G. S. Gordon, A. C. Moses, R. D. Silver, J. S. Flier, and M. C. Carey. Nasal Absorption of Insulin: Enhancement by Hydrophobic Bile Salts. Proc. Natl. Acad. Sci. USA 82:7419–7423 (1985).
A. E. Pontiroli, M. G. Perfetti, B. Fattor, and G. Pozza. Effect of Intranasal Growth Hormone-Releasing Hormone and Corticotropin-Releasing Hormone Administration on Growth Hormone and Cortisol Release: Improved Bioavailability by Means of Sodium-Glycocholate. J. Clin. Endocrinol. Metab. 68:821–824 (1989).
M. Mishima, Y. Wakita, and M. Nakano. Promotion of Nasal Absorption of Insulin by Glycyrrhetinic Acid Derivatives. J. Pharmacobio-Dyn. 12:31–36 (1989).
P. A. Baldwin, C. K. Klingbeil, C. J. Grimm, J. P. Longenecker. The Effect of Sodium Tauro-24,25-Dihydrofusidate on the Nasal Absorption of Human Growth Hormone in Three Animal Models. Pharm. Res. 7:547–552 (1990).
D. J. Pillion, J. D. Bartlett, E. Meezan, M, Yang, R. J. Crain, and W. E. Grizzle. Systemic Absorption of Insulin Delivered Topically to the Rat Eye. Invest. Ophthalmol. Vis. Sci. 32:15–21 (1991).
G. C. Y. Chiou and C. Y. Chuang. Improvement of Systemic Absorption of Insulin through Eyes with Absorption Enhancers. J. Pharm. Sci. 78:815–818 (1989).
C. R. Kensil, U. Patel, M. Lennick, and D. Marciani. Separation and Characterization of Saponins with Adjuvant Activity from Quillaja saponaria Molina Cortex. J. Immunol. 146:431–437 (1991).
C. R. Kensil, S. Soltysik, U. Patel, and D. J. Marciani. Structure/Function Relationship in Adjuvants from Quillaja saponaria Molina. In: R. M. Chanock, H. S. Ginsberg, F. Brown, R. A. Lerner, (eds.), Vaccines 92, Cold Spring Harbor Laboratory Press, Cold Spring, New York, 1992, pp. 35–40.
M. J. Newman, J. Y. Wu, B. H. Gardner, K. J. Monroe, D. Leombruno, J. Recchia, C. R. Kensil, and R. T. Coughlin. Saponin Adjuvant Induction of Ovalbumin Specific CD8+ Cytotoxic T-Lymphocyte Responses. J. Immunol. 148:2357–2362 (1992).
J. Y. Wu, B. H. Gardner, C. I. Murphy, J. Seals, C. R. Kensil, J. Recchia, G. A. Beltz, G. W. Newman, and M. J. Newman. Saponin Adjuvant Enhancement of Antigen-Specific Immune Responses to an Experimental HIV-1 Vaccine. J. Immunol. 148:1519–1525 (1992).
R. Higuchi, Y. Tokimitsu, T. Fujioka, T. Komori, T. Kawasaki, and D. G. Oakenful. Structure of Deacylsaponins Obtained from the Bark of Quillaja Saponaria. Phytochemistry 26:229–235 (1987).
D. J. Pillion, J. Recchia, P. Wang, D. J. Marciani, and C. R. Kensil. DS-1, A Modified Quillaja Saponin, Enhances Ocular and Nasal Absorption of Insulin. J. Pharm. Sci. (In press)
C. R. Kensil, S. Soltysik, and D. J. Marciani. Methods for Enhancing Drug Delivery with Modified Saponins. U.S. Patent Number 5,273,965 (1993).
W. S. York, A. G. Darvil, M. McNeil, T. T. Stevenson, and P. Albersheim. Plant Molecular Biology. In: A. Weissbach and H. Weissbach (eds.), Methods in Enzymology, Academic Press, New York, 1986, volume 118, pp. 3–40.
E. DeVendittis, G. Palumbo, G. Parlato, and V. Bocchini. A Fluorimetric Method for the Estimation of the Critical Micelle Concentration of Surfactants. Anal. Biochem. 115:278–286 (1981).
M. E. Jolley, S. D. Stroupe, C. J. Wang, H. N. Panas, C. L. Keegan, R. L. Schmidt, and K. S. Schwenzer. Fluorescence Polarization Immunoassay. I. Monitoring Aminoglycoside Antibiotics in Serum and Plasma. Clin. Chem. 27:1190–1197 (1981).
C. O. Onyeji, D. P. Nicolau, C. H. Nightingale, R. Quintiliani. Optimal Times above MICs of Ceftibuten and Cefaclor in Experimental Intra-Abdominal Infections. Antimicrobial Agents and Chemotherapy 38:1112–1117 (1994).
G. S. M. J. E. Duchateau, J. Zuidema, and F. W. H. M. Merkus. Bile Salts and Intranasal Drug Absorption. Intl. J. Pharmaceutics 31:193–199 (1986).
A. Rubinstein. Intranasal Administration of Gentamicin in Human Subjects. Antimicrobial Agents and Chemotherapy 23:778–779 (1983).
J. P. Longenecker, A. C. Moses, J. S. Flier, R. D. Silver, M. C. Carey, and E. J. Dubovi. Effects of Sodium Taurodihydrofusidate on Nasal Absorption of Insulin in Sheep. J. Pharm. Sci. 76:351–355 (1987).
D. J. Pillion, C. R. Kensil, J. A. Amsden, and J. Recchia. A Structure/Function Relationship Among Quillaja Saponins Serving as Excipients for Nasal and Ocular Delivery of Insulin. Submitted for publication.
F. W. H. M. Merkus, N. G. M. Schipper, W. A. J. J. Hermens, S. G. Romeijn, and J. C. Verhoef. Absorption Enhancers in Nasal Drug Delivery; Efficacy and Safety. J. Controlled Release 24:201–208 (1993).
S. Gizurarson and E. Bechgaard. Intranasal Administration of Insulin to Humans. Diabetes Res. Clin. Pract. 12:71–84 (1991).
M. T. Scott, M. Goss-Sampson, and R. Bomford. Adjuvant Activity of Saponin: Antigen Localization Studies. Int. Archs. Allergy Appl. Immun. 77:409–412 (1985).
R. Strobbe, G. Charlier, A. van Aert, J. Debecq, and J. Leunen. Studies about the Adjuvant Activity of Saponin Fractions in Foot-and-Mouth Disease Vaccine. II Irritation and Adjuvant Activity of Saponin Fractions Obtained by Chromatography on Sephadex G-100. Arch. Exper. Vet. Med. 28:385–392 (1974).
Rights and permissions
About this article
Cite this article
Recchia, J., Lurantos, M.H.A., Amsden, J.A. et al. A Semisynthetic Quillaja Saponin as a Drug Delivery Agent for Aminoglycoside Antibiotics. Pharm Res 12, 1917–1923 (1995). https://doi.org/10.1023/A:1016235705448
Issue Date:
DOI: https://doi.org/10.1023/A:1016235705448