Abstract
Purpose. To evaluate the chemical/enzymatic stability and the cell permeation characteristics of the modified coumarinic acid-based cyclic prodrug 2 of DADLE (H-Tyr-D-Ala-Gly-Phe-D-Leu-OH), which has an aldehyde equivalent (oxymethyl) inserted between the phenolic group of the promoiety and the carboxylic acid group of the peptide.
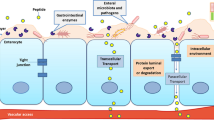
Methods. The rates of the chemical/enzymatic conversion of the oxymethyl-modified prodrug 2 to DADLE were measured by HPLC. The cellular permeation characteristics of DADLE and its oxymethyl-modified prodrug 2 were measured by HPLC using Caco-2 cells, wild type Madin-Darby Canine Kidney cells (MDCK-WT), MDCK cells transfected with human MDR1 gene (MDCK-MDR1), and MDCK cells transfected with human MRP2 gene (MDCK-MRP2) grown onto microporous membranes.
Results. The oxymethyl-modified coumarinic acid-based cyclic prodrug 2 degraded chemically to DADLE in a pH-dependent manner, i.e., rates of conversion increased with increasing pH. The prodrug 2 degraded rapidly in rat plasma (t1/2 = 39 min) and rat liver homogenate (t1/2 = 59.2 min), but much slower in Caco-2 cell homogenate (t1/2 = 678.7 min) and human plasma (t1/2 = 264.3 min). In all four cell lines used for transport studies, the flux rates of the oxymethyl prodrug 2 in the basolateral (BL)-to-apical (AP) direction (Papp BL-to-AP) were significantly greater than the flux rates in the AP-to-BL direction (Papp AP-to-BL). The Papp BL-to-AP /Papp AP-to-BL ratios were >116, 35.1, 21.2, and 12.6 in Caco-2, MDCK-MDR1, MDCK-MRP2, and MDCK-WT cells, respectively. The efflux of the modified prodrug could be inhibited by GF120918 (an inhibitor for P-gp) and cyclosporin A (an inhibitor for P-gp and MRP2).
Conclusions. The oxymethyl-modified coumarinic acid-based cyclic prodrug 2 of DADLE could be converted to DADLE in both chemical and enzymatic media. However, the prodrug was a good substrate for both P-gp and MRP2 suggesting that its permeation across intestinal mucosa and blood-brain barrier would be significantly restricted.
Similar content being viewed by others
REFERENCES
S. Frokjaer, L. Christrup, and P. Krogsgaard-Larson. Peptide and Protein Drug Delivery, Munksgaard, Copenhagen, 1998.
V. H. L. Lee. Peptide and Protein Drug Delivery, Marcel Dekker Inc., New York, 1991.
G. M. Pauletti, S. Gangwar, T. Siahaan, J. Aube, and R. T. Borchardt. Improvement of oral bioavailability: Peptidomimetics and prodrug strategies. Adv. Drug Deliv. Rev. 27:235–256 (1997).
R. Krishnamoorthy and A. K. Mitra. Peptide metabolism by gastric, pancreatic, and lysosomal proteinases. In M. D. Taylor and G. L. Amidon (eds.), Peptide-Based Drug Design: Controlling Transport and Metabolism, American Chemical Society, Washington DC, 1995, pp. 47–68.
J. H. Lin. Human immunodeficiency virus protease inhibitors: from drug design to clinical studies. Adv. Drug Deliv. Rev. 27:215–233 (1997).
M. D. Taylor and G. L. Amidon. Peptide-Based Drug Design: Controlling Transport and Metabolism, American Chemical Society, Washington D.C., 1995.
R. T. Borchardt. Optimizing oral absorption of peptides using prodrug strategies. J. Control. Release 62:231–238 (1999).
W. Wang, R. T. Borchardt, and B. Wang. Orally active peptidommimetics RGD analogue that are glycoprotein IIb/IIIa antagonists. Curr. Med. Chem. 7:437–453 (2000).
S. Gangwar, G. M. Pauletti, T. J. Siahaan, V. J. Stella, and R. T. Borchardt. Synthesis of a novel esterase-sensitive cyclic prodrug of a Hexapeptide using an (acyloxy)alkoxy promoiety. J. Org. Chem. 62:1356–1362 (1997).
B. Wang, S. Gangwar, G. M. Pauletti, T. J. Siahaan, and R. T. Borchardt. Synthesis of a novel esterase-sensitive cyclic prodrug system for peptides that utilizes a 'trimethyl lock'-facilitated lactonization reaction. J. Org. Chem. 62:1363–1367 (1997).
W. Wang, G. Camenisch, D. C. Sane, H. Zhang, E. Hugger, G. L. Wheeler, R. T. Borchardt, and B. Wang. A coumarin-based prodrug strategy to improve the oral absorption of RGD peptidomimetics. J. Control. Release 65:245–251 (2000).
B. Wang, K. Nimkar, W. Wang, H. Zhang, D. Shan, O. Gudmundsson, S. Gangwar, T. J. Siahaan, and R. T. Borchardt. Synthesis and evaluation of the physicochemical properties of esterase-sensitive cyclic prodrugs of opioid peptides using coumarinic acid and phenylpropionic acid linkers. J. Pept. Res. 53:370–382 (1999).
A. Bak, T. J. Siahaan, O. S. Gudmundsson, S. Gangwar, G. J. Friis, and R. T. Borchardt. Synthesis and evaluation of the physicochemical properties of esterase-sensitive cyclic prodrugs of opioid peptides using an (acyloxy)alkoxy linker. J. Pept. Res. 53:393–402 (1999).
O. S. Gudmundsson, K. Nimkar, S. Gangwar, T. J. Siahaan, and R. T. Borchardt. Phenylpropionic acid-based cyclic prodrugs of opioid peptides that exhibit metabolic stability to peptidases and excellent cellular permeation. Pharm. Res. 16:16–23 (1999).
B. Wang, W. Wang, G. P. Camenisch, J. Elmo, H. Zhang, and R. T. Borchardt. Synthesis and evaluation of novel coumarin-based esterase-sensitive cyclic prodrugs of peptidomimetic RGD analogs with improved membrane permeability. Chem. Pharm. Bull. 47:90–95 (1999).
O. S. Gudmundsson, G. M. Pauletti, W. Wang, D. Shan, H. Zhang, B. Wang, and R. T. Borchardt. Coumarinic acid-based cyclic prodrugs of opioid peptides that exhibit metabolic stability to peptidases and excellent cellular permeability. Pharm. Res. 16:7–15 (1999).
A. Bak, O. S. Gudmundsson, G. J. Friis, T. J. Siahaan, and R. T. Borchardt. Acyloxyalkoxy-based cyclic prodrugs of opioid peptides: evaluation of the chemical and enzymatic stability as well as their transport properties across Caco-2 cell monolayers. Pharm. Res. 16:24–29 (1999).
P. Buchwald and N. Bodor. Quantitative structure-metabolism relationships: steric and nonsteric effects in the enzymatic hydrolysis of noncongener carboxylic esters. J. Med. Chem. 42:5160–5168 (1999).
H. Ouyang, T. J. Siahaan, D. Vander Velde, and R. T. Borchardt. Synthesis and conformation study of a coumarinic acid-based cyclic prodrug of opioid peptide with modified sensitivity to esterase-catalyzed bioconversion. J. Peptide Res. 59:183–195 (2002).
J. Gao, E. Hugger, M. Beck-Westermeyer and R. T. Borchardt. Protocols in the application of Caco-2 cells in measuring permeability coefficient of drugs. In A. Doyle, J. B. Griffiths, and D. J. Newell (eds.), Current Protocols in Pharmacology. Vol. 7.2, John Wiley and Sons, Inc., New York, pp. 1–23, 2000.
G. M. Pauletti, S. Gangwar, F. W. Okumu, T. J. Siahaan, V. J. Stella, and R. T. Borchardt. Esterase-sensitive cyclic prodrugs of peptides: evaluation of an acyloxyalkoxy promoiety in a model hexapeptide. Pharm. Res. 13:1615–1623 (1996).
F. Tang and R. T. Borchardt. Characterization of the efflux transporter(s) responsible for restricting intestinal mucosa permeation of the coumarinic acid-based cyclic prodrug of the opioid peptide DADLE. Pharm. Res. 19:787–793 (2002).
F. Tang and R. T. Borchardt. Characterization of the efflux transporter(s) responsible for restricting intestinal mucosa permeation of the acyloxyalkoxy-based cyclic prodrug of the opioid peptide DADLE. Pharm. Res 19:780–786 (2002).
S. P. Letrent, G. M. Pollack, K. R. Brouwer, and K. L. Brouwer. Effects of a potent and specific P-glycoprotein inhibitor on the blood-brain barrier distribution and antinociceptive effect of morphine in the rat. Drug Metab. Dispos. 27:827–834 (1999).
C. Tanaka, R. Kawai, and M. Rowland. Dose-dependent pharmacokinetics of cyclosporin A in rats: events in tissues. Drug Metab. Dispos. 28:582–589 (2000).
A. T. Nies, T. Cantz, M. Brom, I. Leier, and D. Keppler. Expression of the apical conjugate export pump, Mrp2, in the polarized hepatoma cell line, WIF-B. Hepatology 28:1332–1340 (1998).
R. Oliyai. Prodrugs of peptides and peptidomimetics for improved formulation and delivery. Adv. Drug Deliv. Rev. 19:275–286 (1996).
R. A. Cox and K. Yates. An excess acidity analysis of acylal and thioacylal hydrolysis in sulfuric acid. Variation of p with acidity. J. Org. Chem. 51:3619–3624 (1986).
G. M. Pauletti, F. W. Okumu, and R. T. Borchardt. Effect of size and charge on the passive diffusion of peptides across Caco-2 cell monolayers via the paracellular pathway. Pharm. Res. 14:164–168 (1997).
A. H. Dantzig, J. A. Hoskins, L. B. Tabas, S. Bright, R. L. Shepard, I. L. Jenkins, D. C. Duckworth, J. R. Sportsman, D. Mackensen, P. R. Rosteck, Jr., and P. L. Skatrud. Association of intestinal peptide transport with a protein related to the cadherin superfamily. Science 264:430–433 (1994).
S. Matsumoto, H. Saito, and K. Inui. Transcellular transport of Ouyang, Tang, Siahaan, and Borchardt 800 oral cephalosporins in human intestinal epithelial cells, Caco-2: interaction with dipeptide transport systems in apical and basolateral membranes. J. Pharmacol. Exp. Ther. 270:498–504 (1994).
V. J. Wacher, L. Salphati, and L. Z. Benet. Active secretion and enterocytic drug metabolism barriers to drug absorption. Adv. Drug. Deliv. Rev. 46:89–102 (2001).
R. L. Juliano and V. Ling. A surface glycoprotein modulating drug permeability in Chinese hamster ovary cell mutants. Biochim. Biophys. Acta. 455:152–162 (1976).
P. Borst, A. H. Schinkel, J. J. Smit, E. Wagenaar, L. Van Deemter, A. J. Smith, E. W. Eijdems, F. Baas, and G. J. Zaman. Classical and novel forms of multidrug resistance and the physiological functions of P-glycoproteins in mammals. Pharmacol. Ther. 60:289–299 (1993).
C. Cordon-Cardo, J. P. O'Brien, J. Boccia, D. Casals, J. R. Bertino, and M. R. Melamed. Expression of the multidrug resistance gene product (P-glycoprotein) in human normal and tumor tissues. J. Histochem. Cytochem. 38:1277–1287 (1990).
H. Takahashi, R. B. Kim, P. R. Perry, and G. R. Wilkinson. Characterization of the hepatic canalicular membrane transport of a model oligopeptide: ditekiren. J. Pharmacol. Exp. Ther. 281:297–303 (1997).
J. Konig, A. T. Nies, Y. Cui, I. Leier, and D. Keppler. Conjugate export pumps of the multidrug resistance protein (MRP) family: localization, substrate specificity, and MRP2-mediated drug resistance. Biochim. Biophys. Acta 1461:377–394 (1999).
R. Evers, M. Kool, L. van Deemter, H. Janssen, J. Calafat, L. C. Oomen, C. C. Paulusma, R. P. Oude Elferink, F. Baas, A. H. Schinkel, and P. Borst. Drug export activity of the human canalicular multispecific organic anion transporter in polarized kidney MDCK cells expressing cMOAT (MRP2) cDNA. J. Clin. Invest. 101:1310–1319 (1998).
P. Artursson, K. Palm, and K. Luthman. Caco-2 monolayers in experimental and theoretical predictions of drug transport. Adv. Drug Deliv. Rev. 46:27–43 (2001).
H. Gutmann, G. Fricker, M. Torok, S. Michael, C. Beglinger, and J. Drewe. Evidence for different ABC-transporters in Caco-2 cells modulating drug uptake. Pharm. Res. 16:402–407 (1999).
K. I. Hosoya, K. J. Kim, and V. H. Lee. Age-dependent expression of P-glycoprotein gp170 in Caco-2 cell monolayers. Pharm. Res. 13:885–890 (1996).
U. K. Walle, A. Galijatovic, and T. Walle. Transport of the flavonoid chrysin and its conjugated metabolites by the human intestinal cell line Caco-2. Biochem. Pharmacol. 58:431–438 (1999).
A. Braun, S. Hammerle, K. Suda, B. Rothen-Rutishauser, M. Gunthert, S. D. Kramer, and H. Wunderli-Allenspach. Cell cultures as tools in biopharmacy. Eur. J. Pharm. Sci. 11Suppl 2:S51–60 (2000).
I. Pastan, M. M. Gottesman, K. Ueda, E. Lovelace, A. V. Rutherford, and M. C. Willingham. A retrovirus carrying an MDR1 cDNA confers multidrug resistance and polarized expression of P-glycoprotein in MDCK cells. Proc. Natl. Acad. Sci. 85:4486–4490 (1988).
F. Tang and R. T. Borchardt. Are MDCK cells transfected with the human MRP2 gene a good model of the human intestinal mucosa? Pharm. Res. 19:773–779 (2001).
F. Tang and R. T. Borchardt. Are MDCK cells transfected with the human MDR1 gene a good model of the human intestinal mucosa? Pharm. Res. 19:765–772 (2001).
A. J. Smith, U. Mayer, A. H. Schinkel, and P. Borst. Availability of PSC833, a substrate and inhibitor of P-glycoproteins, in various concentrations of serum. J. Natl. Cancer Inst. 90:1161–1166 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ouyang, H., Tang, F., Siahaan, T.J. et al. A Modified Coumarinic Acid-Based Cyclic Prodrug of an Opioid Peptide: Its Enzymatic and Chemical Stability and Cell Permeation Characteristics. Pharm Res 19, 794–801 (2002). https://doi.org/10.1023/A:1016148631055
Issue Date:
DOI: https://doi.org/10.1023/A:1016148631055