Abstract
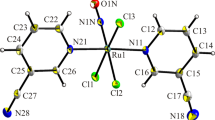
Fine yellowish orange crystals of the binuclear complex K2[RuNO(NO2)3(μ-OH)]2 were obtained by the reaction of K2[RuNO(NO2)4OH] with a stoichiometric amount of 0.8 M nitric acid at room temperature. The compound was investigated by IR spectroscopy, X-ray phase analysis (DRON-3M, CuKα radiation), and X-ray diffraction analysis (Nonius CAD-4, MoKα radiation, graphite monochromator, ω scan mode, 1406 reflections). The crystals are poorly soluble in water and practically insoluble in ethanol and acetone; the compound is stable when stored in air. Crystal data for H2K2N8O16Ru2 are: a = 8.596(2), b = 10.111(1), c = 9.537(1) Å, β = 104.42(1)°, V = 802.8(2) Å3, space group \(P2_1 {\kern 1pt} /{\kern 1pt} c\), Z = 2, ρ calc = 2.691 g/cm3. The structure is built of [RuNO(NO2)3(μ-\(OH)]_2^{2 - } \) complex anions and K+ cations.
Similar content being viewed by others
REFERENCES
A. Joly and M. Vezes, Comp. Rend., 109, 667-670 (1889).
J. M. Fletcher, I. L. Jenkins, F. M. Lever, et al., J. Inorg. Nucl. Chem., 1, No. 2, 378-401 (1955).
S. H. Simonsen and M. H. Muller, ibid., No. 2, 378-401 (1965).
A. J. Blake, R. O. Gould, B. F. J. Johnson, and E. Parisini, Acta Crystallogr. (C), 48, No. 6, 982-984 (1992).
V. N. Pichkov, N. M. Sinitsyn, and O. E. Zvyagintsev, Dokl. Akad. Nauk SSSR, 156, No. 4, 891-893 (1964).
T. S. Khodashova, V. S. Sergienko, and M. A. Porai-Koshits, Zh. Strukt. Khim., 12, No. 3, 478-485 (1971).
N. M. Sinitsyn, V. N. Pichkov, and O. E. Zvyagintsev, Radiokhim., 8, No. 5, 545-555 (1966).
V. N. Pichkov, O. E. Zvyagintsev, and N. M. Sinitsyn, Zh. Neorg. Khim., 11, No. 11, 2560-2568 (1966).
H.-U. Forsterling, ZfI-Mitteilungen, No. 82, 5-85 (1983).
M. A. Fedotov and A. V. Belyaev, Koordinats. Khim., 17, No. 1, 103-111 (1991).
P. G. M. Brown, J. Inorg. Nucl. Chem., 13, No. 1, 73-83 (1960).
V. A. Emel'yanov, M. A. Fedotov, and A. V. Belyaev, Zh. Neorg. Khim., 37, No. 12, 2717-2721 (1992).
V. A. Emel'yanov, A. V. Belyaev, M. A. Fedotov, et al., ibid., 37, No. 11, 2515-2523 (1992).
L. A. Butman, T. S. Khodashova, L. Kh. Minacheva, and V. I. Tayukin, Zh. Strukt. Khim., 5, No. 2, 250-256 (1964).
E. E. Mercer, W. A. Mccallister, and J. R. Durig, Inorg. Chem., 5, No. 11, 1881-1886 (1966).
G. M. Sheldrick, SHELX-97, Release 97-2, University of Göttingen, Germany (1998).
A. Svetlanova-Larsen, C. R. Zoch, and J. L. Hubbard, Organometallics, 15, 3076 (1996).
S. V. Borisov, Zh. Strukt. Khim., 27, No. 3, 164-166 (1986).
N. A. Bliznyuk and S. V. Borisov, “Calculation of atomic reticular density, regularity, and similarity of atomic matrices: algorithms, programs, examples,” preprint No. 89-14, Institute of Catalysis, Siberian Branch, Russian Academy of Sciences, Novosibirsk (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Emelyanov, V.A., Gromilov, S.A., Baidina, I.A. et al. Formation of a Dimer with a Double Hydroxo Bridge in Nitric Acid Solutions of the Nitrosotetranitrohydroxo Complex of Ruthenium(II). Crystal Structure of K2[RuNO(NO2)3(μ-OH)]2. Journal of Structural Chemistry 43, 147–152 (2002). https://doi.org/10.1023/A:1016042404186
Issue Date:
DOI: https://doi.org/10.1023/A:1016042404186