Abstract
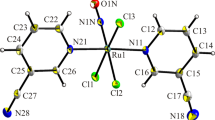
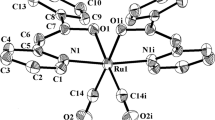
A reaction of nitric acid with [RuNO(Py)2(NO2)2OH] without heating is shown to result in the complete substitution of nitro and hydroxo ligands with the formation of [RuNO(Py)2(NO3)3] (1). The high lability of nitrate ligands allows their subsequent substitution with the formation of [RuNO(Py)3(NO3)(OH)](NO3) complex (2). The compounds are characterized by the elemental analysis and IR spectroscopy. Their crystal structures are determined by X-ray crystallography. The crystallographic data for complex 1 are: a = 8.9174(4) Å, b = 12.2822(5) Å, c = 15.9104(6) Å, β = 94.4160(10)°, space group P21/c, Z = 4, R = 0.0468 [I > 2σ(I)]; for complex 2: a = 10.8147(4) Å, b = 12.6004(5) Å, c = 14.2201(5) Å, β = 101.5750(10)°, space group Cc, Z = 4, R = 0.0174. Complex 1 is obtained as a facial isomer. Oxygen atoms of coordinated nitrate ligands occupy one of the faces of a distorted RuN3O3 octahedron. In complex 2, the hydroxo ligand is coordinated in the trans-position to the nitroso group. The equatorial positions in the octahedral ruthenium environment are occupied by three nitrogen atoms of pyridine ligands and the oxygen atom of the nitrate ligand.
Similar content being viewed by others
References
P. Coppens, I. Novozhilova, and A. Kovalevsky. Chem. Rev., 2002, 102, 861–884.
A. A. Mikhailov, E. Wenger, G. A. Kostin, and D. Schaniel. Chem. — A Eur. J., 2019, 25, 7569–7574.
G. A. Kostin, V. Vorob’yev, A. A. Mikhailov, and N. V. Kuratieva. J. Mol. Struct., 2019, 1193, 334–341.
N. Casaretto, B. Fournier, S. Pillet, E. E. Bendeif, D. Schaniel, A. K. E. Gallien, P. Klüfers, and T. Woike. CrystEngComm, 2016, 18, 7260–7268.
J. Akl, C. Billot, P. G. Lacroix, I. Sasaki, S. Mallet-Ladeira, I. Malfant, R. Arcos-Ramos, M. Romero, and N. Farfán. New J. Chem., 2013, 37, 3518–3527.
R. B. Morgunov, A. I. Dmitriev, F. B. Mushenok, É. B. Yagubskiĭ, L. A. Kushch, A. R. Mustafina, V. A. Burilov, A. T. Gubaĭdullin, A. I. Konovalov, I. S. Antipin, and Y. Tanimoto. Phys. Solid State, 2009, 51, 2095–2100.
G. A. Kostin, A. O. Borodin, N. V. Kuratieva, A. S. Bogomyakov, and A. A. Mikhailov. Inorg. Chim. Acta, 2018, 479, 135–140.
A. A. Mikhailov, V. A. Vorob’yev, V. A. Nadolinny, Y. V. Patrushev, Y. S. Yudina, and G. A. Kostin. J. Photochem. Photobiol. A Chem., 2019, 373, 37–44.
G. A. Kostin, A. A. Mikhailov, N. V. Kuratieva, D. P. Pischur, D. O. Zharkov, and I. R. Grin. New J. Chem., 2017, 41, 7758–7765.
E. Tfouni, D. R. Truzzi, A. Tavares, A. J. Gomes, L. E. Figueiredo, and D. W. Franco. Nitric Oxide, 2012, 26, 38–53.
J. M. Fletcher, I. L. Jenkins, F. M. Lever, F. S. Martin, A. R. Powell, and R. Todd. J. Inorg. Nucl. Chem., 1955, 1, 378–401.
E. V. Kabin, V. A. Emel’yanov, and S. V. Tkachev. Russ. J. Inorg. Chem., 2013, 58, 229–238.
V. A. Emel’yanov and M. A. Fedotov. Russ. J. Inorg. Chem., 2006, 51, 1811–1819.
B. Coe, C. I. McDonald, and R. L. Beddoes. Polyhedron, 1998, 17, 1997–2007.
M. Mukaida, Y. Sato, H. Kato, M. Mori, D. Ooyama, H. Nagao, and F. S. Howell. Bull. Chem. Soc. Jpn., 2000, 73, 85–95.
M. A. Il’in, E. V. Kabin, V. A. Emel’yanov, I. A. Baidina, and V. A. Vorob’yov. J. Struct. Chem., 2009, 50, 328–334.
V. A. Vorobyev, V. A. Emelyanov, I. A. Baidina, and D. A. Piryazev. J. Struct. Chem., 2017, 58, 975–982.
G. A. Kostin, A. O. Borodin, A. A. Mikhailov, N. V. Kuratieva, B. A. Kolesov, D. P. Pishchur, T. Woike, and D. Schaniel. Eur. J. Inorg. Chem., 2015, 2015, 4905–4913.
G. A. Kostin, A. A. Mikhailov, N. V. Kuratieva, S. V. Tkachev, D. Schaniel, and T. Woike. Eur. J. Inorg. Chem., 2016, 2016, 4045–4053.
A. B. P. Lever, E. Mantovani, and B. S. Ramaswamy. Can. J. Chem., 1971, 49, 1957–1964.
G. M. Sheldrick. Acta Crystallogr., Sect. A, 2015, 71, 3–8.
A. N. Makhinya, M. A. Il’In, E. V. Kabin, I. A. Baidina, M. R. Gallyamov, and N. I. Alferova. Russ. J. Coord. Chem., 2014, 40, 297–303.
A. N. Makhinya, M. A. Il’In, I. A. Baidina, P. E. Plyusnin, and M. R. Gallyamov. J. Struct. Chem., 2014, 55, 682–690.
A. O. Borodin, G. A. Kostin, P. E. Plusnin, E. Y. Filatov, A. S. Bogomyakov, and N. V. Kuratieva. Eur. J. Inorg. Chem., 2012, 2012, 2298–2304.
A. N. Makhinya, M. A. Il’In, I. A. Baidina, P. E. Plusnin, N. I. Alferova, and D. P. Pishchur. J. Struct. Chem., 2014, 55, 311–318.
O. A. Plyusnina, V. A. Emel’yanov, I. A. Baidina, P. E. Plyusnin, and S. A. Gromilov. J. Struct. Chem., 2011, 52, 140–150.
Funding
The work was supported by RFBR grant No. 19-03-00594.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interests
The authors declare that they have no conflict of interests.
Russian Text © The Author(s), 2020, published in Zhurnal Strukturnoi Khimii, 2020, Vol. 61, No. 1, pp. 91–99.
Rights and permissions
About this article
Cite this article
Kostin, G.A., Nikiforov, Y.A. & Kuratieva, N.V. Synthesis and Structure of Ruthenium Nitroso Complexes with Nitrate Anions and Pyridine as Ligands. J Struct Chem 61, 86–94 (2020). https://doi.org/10.1134/S0022476620010096
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476620010096