Abstract
Purpose. This work was performed in order to compare and contrast results obtained from cascade impactor measurements on metered dose inhalers (MDIs) using a variety of inlet ports.
Methods. The collection characteristics of four cascade impactor ports (a physical model of the human throat, a simplified geometry intended to mimic the physical model, and two currently-used ports) were measured on a variety of MDI formulations.
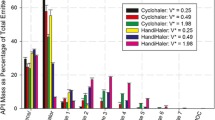
Results. The portion of the MDI spray which collects on the entry port depends in a complicated fashion on the characteristics of the formulation; in these studies the fraction of the total dose which was collected on the port ranged between about 20% and 90% of the total emitted dose. The collection characteristics of the simplified geometry closely corresponded to the physical model. The length of the flow path between the port and the impactor was varied, and found not to have a strong effect on the measured size distribution passing the port.
Conclusions. Ranking of various MDI formulations according to performance criteria as measured with a cascade impactor should be expected to depend on the particular inlet port which is used.
Similar content being viewed by others
REFERENCES
A. R. Clark. The physics of aerosol formulation by MDIs—limitations of the current approach. J. Biopharm Sci 3(1/2): 69–76 (1992).
R. N. Dalby and S. L. Tiano. Pitfalls and opportunities in the inertial sizing and output testing of nebulizers. Pharm Tech Sept:144–56 (1993).
K. Fults, T. D. Cyr, and A. J. Hickey. The influence of sampling chamber dimensions on aerosol particle size measurement by cascade impactor and twin impinger. J Pharm Pharmacol 43:726–728 (1991).
W. A. Hunke and A. B. C. Yu. Particle-size distribution of bitolterol mesylate solution delivered by four compressed-air nebulizer devices. Am J Hosp Pharm 44:1392–1396 (1987).
C. S. Kim, A. Eldridge, and M. Sackner. Oropharyngeal deposition and delivery aspects of metered-dose inhaler aerosols. Am Rev Respir Dis 135:157–164 (1987).
G. P. Martin, A. E. Bell, and C. Marriott. An in vitro method for assessing particle deposition form metered pressurized aerosols and dry powder inhalers. Int J Pharm 44:57–63 (1988).
R. W. Niven, R. M. Kacmarek, J. D. Brain, and R. A. Peterfreund. Small bore nozzle extensions to improve the delivery efficiency of drugs from metered dose inhalers: laboratory evaluation. Ann Rev Respir Dis 147:1590–1594 (1993).
J. J. Sciarra and A. Cutie. Simulated respiratory system for in vitro evaluation of two inhalation delivery systems using selected steroids. J. Pharm Sci 67(10):1428–1431 (1978).
M. Van Oort, R. O. Gollmar, and R. J. Bohinski. Effects of sampling chamber volume and geometry on aerodynamic size distributions of metered-dose inhalation aerosols measured with the Andersen cascade impactor. Pharm Res 11(4):604–607 (1994).
D. J. Velasquez, Personal communication. (1991).
Aerosols [601] USP XXIII, 1764. The United States Pharmacopeial Convention, Inc. (1994).
D. L. Swift. The oral airway—a conduit or collector for pharmaceutical aerosols? in Respiratory Drug Delivery IV, P. R. Byron, R. N. Dalby, and S. J. Farr, eds. Interpharm Press. (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Miller, N.C., Purrington, A.M. A Cascade Impactor Entry Port for MDI Sprays with Collection Characteristics Imitating a Physical Model of the Human Throat. Pharm Res 13, 391–397 (1996). https://doi.org/10.1023/A:1016040509063
Issue Date:
DOI: https://doi.org/10.1023/A:1016040509063