Abstract
Purpose. Herein we report the discovery of a group of derivatized α-amino acids that increase the oral bioavailability of sodium cromolyn.
Methods. We prepared three N-acylated α-amino acids and used these compounds to demonstrate the oral delivery of cromolyn in an in vivo rat model. In vitro experiments, including permeation studies and near infrared spectroscopy, were also performed to initiate an understanding of the mechanism by which these compounds facilitate cromolyn oral delivery.
Results. Following oral administration to rats of solutions containing a combination of cromolyn and the delivery agent, significant systemic plasma concentrations of the drug were detected. In vitro studies suggest that absorption of the drug across the gastrointestinal membrane is a passive process.
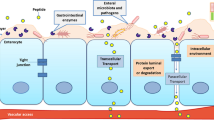
Conclusions. The absolute oral bioavailability of sodium cromolyn in the rat model is estimated to be ~5%. Preliminary mechanistic studies suggest that a complex of the cromolyn/delivery agent facilitates permeation across/through the membrane.
Similar content being viewed by others
REFERENCES
G. G. Shapiro and P. Konig. Cromolyn sodium: a review, Pharmacotherapy 5:156–170 (1985).
T. W. Rall. Sodium cromolyn. In A. G. Gilman, T. W. Rall, A. S. Nies, and P. Taylor (eds), The Pharmacological Basis of Therapeutics, 8th ed., Macmillan, New York, 1990, pp 630–635.
A. M. Edwards. Sodium cromoglycate (Intal®) as an anti-inflammatory agent for the treatment of chronic asthma, Clin. Exptl. Allergy 24:612–623 (1994).
A. Yoshimi, H. Hashizume, M. Kitagawa, K. Nishimura, and N. Kakeya. Absorption mechanism of 1,3-bis-(2-ethoxycarbonylchromon-5-yloxy)-2-((S)-lysyloxy)propane dihydrochloride (N-556), a prodrug for the oral delivery of disodium cromoglycate, Biol. Pharm. Bull. 16:375–378 (1993) references cited therein.
L. S. Schanker, D. J. Tocco, B. B. Brodie, and C. A. M. Hogben. Absorption of drugs from the rat samll intestine, J. Pharmacol. Exp. Ther. 123:81–88 (1958).
C. A. M. Hogben, D. J. Tocco, B. B. Brodie, and L. S. Schanker. On the mechanism of intestinal absorption of drugs, J. Pharmacol. Exp. Ther. 125:275–282 (1959).
G. F. Moss, K. M. Jones, J. T. Ritchie, and J. S. G. Cox. Distribution and metabolism of disodium cromoglycate in rats, Toxicol. Appl. Pharmacol. 17:691–698 (1970).
S. K. Gupta, S. Kumar, S. Bolton, C. R. Behl, and A. W. Malick. Optimization of iontophoretic transdermal delivery of a peptide and non-peptide drug, J. Cont. Rel. 30:253–261 (1994).
G. F. Moss, K. M. Jones, J. T. Ritchie, and J. S. G. Cox. Plasma levels and urinary excretion of disodium cromoglycate after inhalation by human volunteers, Tox. Appl. Pharm. 20:147–156 (1971).
M. Kaartinen, A. Pentilla, and P. T. Kovanen. Mast cells of two types differing in neutral protease composition in the human arotic intima, Arterioscler. Thromb. 14:966–972 (1994).
T. Mori, K. Nishimura, S. Tamaki, S. Nakamura, H. Tsuda, and N. Kakaya. Prodrugs for the oral delivery of disodium cromoglycate, Chem. Pharm. Bull. 36:338–344 (1988).
A. Yoshimi, H. Hashizume, M. Kitagawa, K. Nishimura, and N. Kakeya. Characteristics of 1,3-bis-(2-ethoxycarbonylchromon-5-yloxy)-2-((S)-lysyloxy)propane dihydrochloride (N-556), a prodrug for the oral delivery of disodium cromoglycate in absorption and excretion in rats and rabbits, J. Pharmacobio-Dyn. 15:681–686 (1992).
G. Patonay, M. D. Antoine, D. Srinivasan, and L. Strekowski. Near-infrared probe for determination of solvent hydrophobicity, Applied Spectroscopy 45:457–460 (1991).
A. Leone-Bay, C. Mcinnes, N. Wang, F. DeMorin, D. Achan, C. Lercara, D. Sarubbi, S. Haas, J. Press, E. Barantsevich, B. O'Broin, S. Milstein and D. R. Paton. Microsphere formation in a series of derivatized α-amino acids: properties, moledular modeling, and oral delivery of salmon calcitonin, J. Med. Chem. 38:4257–4261 (1995).
A. Leone-Bay, N. Santiago, D. Achan, K. Chaudhary, F. DeMorin, L. Falzarano, S. Haas, S. Kalbag, D. Kaplan, H. Leipold, C. Lercara, D. O'Toole, T. Rivera, C. Rosado, D. Sarubbi, E. Vuocolo, N. Wang, S. Milstein, and R. A. Baughman. N-Acylated α-amino acids as novel oral delivery agents for proteins, J. Med. Chem. 38:4262–4269 (1995).
J. Kobayashi and Y. Machida. Studies on absorption, excretion and tolerance of an oral form of disodium cromoglycate (DSCG) in healthy volunteers, Jpn. Pharmacol. Ther. 8:80–91 (1980).
M. Gibaldi, and D. Perrier. Pharmacokinetics, 2nd ed.; Marcel Dekker: New York, 1982; pp 169–180.
H. H. Ussing, and K. Zerahn. Active transport of sodium as the source of electric current in the short-circuited isolated frog skin, Acta Phys. Scand. 23:110–133 (1951).
S. C. Sutton, A. E. Forbes, R. Cargill, J. H. Hochman, and E. L. LeCluyse. Simultaneous in vitro measurement of intestinal tissue permeability and transepithelial electrical resistance (TEER) using Sweetana-Grass diffusion cells, Pharm. Res. 9:316–319 (1992).
B. Yao, D. L. Hogan, K. Bukhave, M. A. Koss, and J. I. Isenberg. Bicarbonate transport by rabbit duodenum in vitro: effect of vasoactive intestinal polypeptide, prostaglandin E2, and cyclic adenosine monophosphate, Gastroenterology 104:732–740 (1993).
T. R. Kleyman, E, J. Cragoe. Amiloride and its analogs as tools in the study of ion transport, J. Membrane Biol. 105:1–21 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Leone-Bay, A., Leipold, H., Sarubbi, D. et al. Oral Delivery of Sodium Cromolyn: Preliminary Studies In Vivo and In Vitro . Pharm Res 13, 222–226 (1996). https://doi.org/10.1023/A:1016034913181
Issue Date:
DOI: https://doi.org/10.1023/A:1016034913181