Abstract
Iron and copper play major roles in biological systems, catalyzing free radical production and consequently causing damage. The relatively high levels of these metals, which are mobilized into the coronary flow following prolonged ischemia, have been incriminated as key players in reperfusion injury to the heart. In the present communication we investigated other roles of iron – providing protection to the ischemic heart via preconditioning (PC).
PC was accomplished by subjecting isolated rat hearts to three episodes of 2 min ischemia separated by 3 min of reperfusion. Prolonged ischemia followed the PC phase. PC hearts (group I) were compared to hearts subjected to normal perfusion (group II, no ischemia) and to ischemia without PC (group III). Group I showed a marked improvement in the recovery of hemodynamic function vs. group III. Biochemical parameters further substantiated the PC protection provided to group I against prolonged ischemia. Correspondingly, group I presented markedly lower re-distribution and mobilization of iron and copper into the coronary flow, following prolonged ischemia, as evinced from the decrease in total levels, and in the 'free' fraction of iron and copper.
During the PC phase no loss of cardiac function was observed. A small wave of re-distribution and mobilization of iron (typically less than 4–8% of the value of 35 min ischemia) was recorded. The cellular content of ferritin (Ft) measured in the heart was significantly higher in group I than in group III (0.90 and 0.54 μg/mg, respectively). Also, iron-saturation of Ft was significantly lower for PC hearts, compared to both groups II and III (0.22 vs. 0.32 and 0.31 μg/mg, for 35 min ischemia, respectively). These findings are in accord with the proposal that intracellular re-distribution and mobilization of small levels of iron, during PC, cause rapid accumulation of ferritin – the major iron-storage protein.
It is proposed that iron play a dual role: (i) It serves as a signaling pathway for the accumulation of Ft following the PC phase. This iron is not involved in cardiac injury, but rather prepares the heart against future high levels of 'free' iron, thus reducing the degree of myocardial damage after prolonged ischemia. (ii) High levels of iron (and copper) are mobilized following prolonged ischemia and cause tissue damage.
Similar content being viewed by others
References
De Jong JW, de Jonge R, Keijzer E, Bradamante S: The role of adenosine in preconditioning. Pharmacol Ther 87: 141–149, 2000
Nakano A, Cohen MV, Downey JM: Ischemic preconditioning: From basic mechanisms to clinical applications. Pharmacol Ther 86: 263–275, 2000
Tomai F, Crea F, Chiariello L, Gioffre PA: Ischemic preconditioning in humans: Models, mediators, and clinical relevance. Circulation 100: 559–563, 1999
Das DK, Engelman RM, Maulik N: Oxygen free radical signaling in ischemic preconditioning. Ann NY Acad Sci 30: 49–65, 1999
Przyklenk K, Simkhovich BZ, Bauer B: Cellular mechanisms of infarct size reduction with ischemic preconditioning. Role of calcium? Ann NY Acad Sci 30: 192–210, 1999
Yue Y, Krenz M, Cohen MV, Downey JM, Critz SD: Menadione mimics the infarct-limiting effect of preconditioning in isolated rat hearts. Am J Physiol Heart Circ Physiol 281: H590–H595, 2001
DeBoer DA, Clark RE: Iron chelation in myocardial preservation after ischemia-reperfusion injury: The importance of pretreatment and toxicity. Ann Thorac Surg 53: 412–418, 1992
Zeltcer G, Berenshtein E, Samuni A, Chevion M: Nitroxide radicals prevent metal-aggravated reperfusion injury in isolated rat heart. Free Radic Res Commun 27: 627–635, 1997
Chevion M, Jiang Y, Har El R: Copper and iron are mobilized following myocardial ischemia: Possible predictive criteria for tissue injury. Proc Natl Acad Sci USA 90: 1102–1106, 1993
Berenshtein E, Mayer B, Goldberg C, Kitrossky N, Chevion M: Patterns of mobilization of copper and iron following myocardial ischemia: Pos-291 sible predictive criteria for tissue injury. J Mol Cell Cardiol 29: 3025–3034, 1997
Cottin Y, Doise JM, Maupoil V: Plasma iron status and lipid peroxidation following thrombolytic therapy for acute myocardial infarction. Fund Clin Pharmacol 12: 236–241, 1998
Eaton S, Pourfarzam M, Bartlett K: The effect of respiratory chain impairment of beta-oxidation in rat heart mitochondria. Biochem J 319: 633–640, 1996
White K, Munro HN: Induction of ferritin subunit synthesis by iron is regulated at both the transcriptional and translational levels. J Biol Chem 263: 8938–8942, 1988
van der Kraaij AM, van Eijk HG, Koster JF: Prevention of postischemic cardiac injury by the orally active iron chelator 1,2-dimethyl-3-hydroxy-4-pyridone (L1) and the antioxidant (+)-cyanidanol-3. Circulation 80: 158–164, 1989
Lesnefsky EJ, Ye J: Exogenous intracellular, but not extracellular, iron augments myocardial reperfusion injury. Am J Physiol 266: H384–H392, 1994
Biemond P, Swaak AJ, van Eijk HG, Koster JF: Superoxide dependent iron release from ferritin in inflammatory diseases. Free Radic Biol Med 4: 185–198, 1988
Allen DR, Wallis GL, McCay PB: Catechol adrenergic agents enhance hydroxyl radical generation in xanthine oxidase systems containing ferritin: Implications for ischemia/reperfusion. Arch Biochem Biophys 315: 235–243, 1994
Bando Y, Aki K: Superoxide-mediated release of iron from ferritin by some flavoenzymes. Biochem Biophys Res Commun 168: 389–395, 1990
Vaisman B, Fibach E, Konijn AM: Utilization of intracellular ferritin iron for hemoglobin synthesis in developing human erythroid precursors. Blood 90: 831–838, 1997
Sakaida I, Kyle ME, Farber JL: Autophagic degradation of protein generates a pool of ferric iron required for the killing of cultured hepatocytes by an oxidative stress. Mol Pharmacol 37: 435–442, 1990
Rothman RJ, Serroni A, Farber JL: Cellular pool of transient ferric iron, chelatable by deferoxamine and distinct from ferritin, that is involved in oxidative cell injury. Mol Pharmacol 42: 703–710, 1992
Roberts S, Bomford A: Ferritin iron kinetics and protein turnover in K562 cells. J Biol Chem 263: 19181–19187, 1988
Langendorff O: Untersuchungen am uberlebenden Saugethierherzen. Pflügers Arch 61: 291–332, 1895
Rothery E: Varian analytical methods for graphite tube atomizers. Sugar Land, TX, Varian, 1988
Bradford MM: A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248–254, 1976
Konijn AM, Tal R, Levy R, Matzner Y: Isolation and fractionation of ferritin from human term placenta - a source for human isoferritins. Anal Biochem 144: 423–428, 1985
Konijn AM, Levy R, Link G, Hershko C: A rapid and sensitive ELISA for serum ferritin employing a fluorogenic substrate. J Immunol Meth 54: 297–307, 1982
Udassin R, Ariel I, Haskel Y, Kitrossky N, Chevion M: Salicylate as an in vivo free radical trap: Studies on ischemic insult to the rat intestine. Free Radic Biol Med 10: 1–6, 1991
Halliwell B, Grootveld M, Gutteridge JM: Methods for the measurement of hydroxyl radicals in biomedical systems: deoxyribose degradation and aromatic hydroxylation. Meth Biochem Anal 33: 59–90, 1988
Harrison PM, Arosio P: The ferritins: Molecular properties, iron storage function and cellular regulation. Biochim Biophys Acta 1275: 161–203, 1996
Picard V, Renaudie F, Porcher C: Overexpression of the ferritin H subunit in cultured erythroid cells changes the intracellular iron distribution. Blood 87: 2057–2064, 1996
Picard V, Epsztejn S, Santambrogio P, Cabantchik ZI, Beaumont C: Role of ferritin in the control of the labile iron pool in murine erythroleukemia cells. J Biol Chem 273: 15382–15386, 1998
Konijn AM, Glickstein H, Vaisman B: The cellular labile iron pool and intracellular ferritin in K562 cells. Blood 94: 2128–2134, 1999
Karwatowska-Prokopczuk E, Czarnowska E, Beresewicz A: Iron availability and free radical induced injury in the isolated ischaemic/reperfused rat heart. Cardiovasc Res 26: 58–66, 1992
Balla G, Jacob HS, Balla J: Ferritin: A cytoprotective antioxidant strategem of endothelium. J Biol Chem 267: 18148–18153, 1992
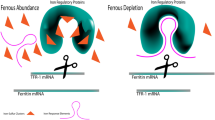
Theil EC: Iron regulatory elements (IREs): A family of mRNA noncoding sequences. Biochem J 304: 1–11, 1994
Klausner RD, Rouault TA, Harford JB: Regulating the fate of mRNA: The control of cellular iron metabolism. Cell 72: 19–28, 1993
Hentze MW, Kuhn LC: Molecular control of vertebrate iron metabolism: mRNA-based regulatory circuits operated by iron, nitric oxide, and oxidative stress. Proc Natl Acad Sci USA 93: 8175–8182, 1996
Roberts S, Bomford A: Chelation of transferrin iron by desferrioxamine in K562 cells. The partition of iron between ferrioxamine and ferritin. Biochem J 254: 869–875, 1988
Shibata T, Yamamoto F, Suehiro S, Kinoshita H: Effects of protease inhibitors on postischemic recovery of the heart. Cardiovasc Drugs Ther 11: 547–556, 1997
Karapetian RG, Oganesian SS, Akopian TN, Dzhanpoladian EG: Activity of proteolytic enzymes and their inhibitors in experimental myocardial ischemia. Vopr Med Khim 39: 29–31, 1993
Chevion M: A site-specific mechanism for free radical induced biological damage: The essential role of redox-active transition metals. Free Radic Biol Med 5: 27–37, 1988
Gelvan D, Saltman P, Powell SR: Cardiac reperfusion damage prevented by a nitroxide free radical. Proc Natl Acad Sci USA 88: 4680–4684, 1991
Ambrosio G, Flaherty JT, Duilio C: Oxygen radicals generated at reflow induce peroxidation of membrane lipids in reperfused hearts. J Clin Invest 87: 2056–2066, 1991
Cairo G, Tacchini L, Pogliaghi G: Induction of ferritin synthesis by oxidative stress. Transcriptional and post-transcriptional regulation by expansion of the 'free' iron pool. J Biol Chem 270: 700–703, 1995
Balla J, Jacob HS, Balla G: Endothelial-cell heme uptake from heme proteins: induction of sensitization and desensitization to oxidant damage. Proc Natl Acad Sci USA 90: 9285–9289, 1993
Vile GF, Basu-Modak S, Waltner C, Tyrrell RM: Heme oxygenase 1 mediates an adaptive response to oxidative stress in human skin fibroblasts. Proc Natl Acad Sci USA 91: 2607–10, 1994
Vile GF, Tyrrell RM: Oxidative stress resulting from ultraviolet A irradiation of human skin fibroblasts leads to a heme oxygenase-dependent increase in ferritin. J Biol Chem 268: 14678–14681, 1993
Toth I, Yuan L, Rogers JT, Boyce H, Bridges KR: Hypoxia alters ironregulatory protein-1 binding capacity and modulates cellular iron homeostasis in human hepatoma and erythroleukemia cells. J Biol Chem 274: 4467–4473, 1999
Thornton J, Striplin S, Liu GS: Inhibition of protein synthesis does not block myocardial protection afforded by preconditioning. Am J Physiol 259: H1822–H1825, 1990
Rowland RT, Meng X, Cleveland JC: Cardioadaptation induced by cyclic ischemic preconditioning is mediated by translational regulation of de novo protein synthesis. J Surg Res 71: 155–160, 1997
Matsuyama N, Leavens JE, McKinnon D: Ischemic but not pharmacological preconditioning requires protein synthesis. Circulation 102: III312–III318, 2000
Emerson MR, Samson FE, Pazdernik TL: Effects of hypoxia preconditioning on expression of metallothionein-1,2 and heme oxygenase-1 before and after kainic acid-induced seizures. Cell Mol Biol (Noisyle-grand) 46: 619–626, 2000
Chen K, Zhang J, Ye C: Metallothionein involvement in the delayed protection after ischemic or anoxic preconditioning in myocardium or cultured cardiomyocytes. Zhonghua Yi Xue Za Zhi 77: 106–110, 1997
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Berenshtein, E., Vaisman, B., Goldberg-Langerman, C. et al. Roles of ferritin and iron in ischemic preconditioning of the heart. Mol Cell Biochem 234, 283–292 (2002). https://doi.org/10.1023/A:1015923202082
Issue Date:
DOI: https://doi.org/10.1023/A:1015923202082