Abstract
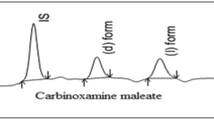
A method was developed and validated for the simultaneous analysis of (+)- and (−)-bevantolol in human plasma. The assay involves plasma protein precipitation, derivatization of racemic bevantolol to its diastereomeric thioureas with 2,3,4,5-tetra-o-acetyl-α-D-gluco-pyranosyl isothiocyanate, and solid-phase extraction of the diastereomers from 0.5 ml human plasma. Chromatographic separation was accomplished under isocratic conditions using a reversed-phase C-18 analytical column and mobile phase consisting of equal parts of 75 mM dibasic ammonium phosphate buffer (adjusted to pH 3.5 with phosphoric acid) and acetonitrile, with a detection wavelength of 220 nm. The absolute peak-height method was employed for quantitation. Retention times for the diastereomers of ( + )- and (−)-bevantolol were 7.4 and 6.4 min, respectively. The method is suitable for the quantification of the enantiomers over a concentration range of 40 to 800 ng/ml per enantiomer.
Similar content being viewed by others
REFERENCES
A. M. Barrett and V. A. Cullum. The biological properties of the optical isomers of propranolol and their effects on cardiac arrhythmias. Br. J. Pharmacol. 13:43–55 (1968).
M. S. Lennard, G. T. Tucker, J. H. Silas, S. Freestone, L. E. Ramsay, and H. F. Woods. Differential stereoselective metabolism of metoprolol in extensive and poor debrisoquin metabolizers. Clin. Pharmacol. Ther. 34:732–737 (1983).
P. H. Hsyu and K. M. Giacomini. Stereoselective renal clearance of pindolol in humans. J. din. Invest. 76:1720–1726 (1985).
U. K. Walle, T. Walle, S. A. Bai, and L. S. Olanoff. Stereoselective binding of propranolol to human plasma, α1-acid glycoprotein, and albumin. Clin. Pharmacol. Ther. 34:718–723 (1983).
Internal communication, Warner-Lambert Co., 1974.
L. Olanoff, T. Walle, K. Walle, T. D. Coward, and T. E. Gaffney. Stereoselective clearance and distribution of intravenous propranolol. Clin. Pharmacol. Ther. 35:755–762 (1985).
T. Kinoshita, Y. Kasahara, and N. Nimura. Reversed-phase high-performance liquid chromatographic resolution of non-esterified enantiomeric amino acids by derivatization with 2,3,4,5-tetra-o-acetyl-β-D-glucopyranosyl isothiocyanate and 2,3,4-tri-o-acetyl-α-D-arabinopyranosyl isothiocyanate. J. Chromatogr. 210:77–82 (1981).
M. Hoefle, S. Hastings, R. Meyer, R. Corey, A. Holmes, and C. Stratton. Cardioselective β-adrenergic blocking agents. 1. 1-[(3,4-Dimethoxyphenethyl)amino]-3-aryloxy-2-propanols. J. Med. Chem. 18:148–152 (1975).
B. Silber and S. Riegelman. Stereospecific assay for (−)-and (+)-propranolol in human and dog plasma. J. Pharmacol. Exp. Ther. 215:643–648 (1980).
T. Walle and U. Walle. Stereoselective oral bioavailability of (±)-propranolol in the dog. A GC-MS study using a stable isotope technique. Res. Commun. Chem. Pathol. Pharmacol. 23:453–464 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rose, S.E., Randinitis, E.J. A High-Performance Liquid Chromatographic Assay for the Enantiomers of Bevantolol in Human Plasma. Pharm Res 8, 758–762 (1991). https://doi.org/10.1023/A:1015858219018
Issue Date:
DOI: https://doi.org/10.1023/A:1015858219018