Abstract
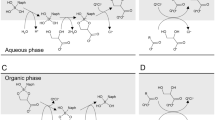
A general mechanism for oxidizing reducing sugars (pentoses, hexoses and disaccharides) by OsO4 in alkaline medium is proposed. The reactions exhibit pseudounimolecular kinetics with respect to OsO4, are first order with respect to lower [sugar] and [OH−], but tend towards zeroth order with respect to both higher [sugar] and [OH−]. These results suggest the formation of an activated complex between the enediol andd osmium tetroxide, which slowly disproportionates to give osmium(VI) species and the intermediate products. The reaction products were identified. When an aldehydo sugar is treated with alkali, structural changes occur and an equilibrium is set up, mainly involving a keto sugar product and the starting aldehydo sugar consumed. This process is reversible. These changes are mainly due to the well-known Lobry-de-Bruyn-Alberda Van Ekenstein transformations.
Similar content being viewed by others
References
H. S. Singh, O. C. Saxena and M. P. Singh, J. Am. Chem. Soc., 92, 537 (1970).
N. Nath and M. P. Singh, Phys. Chem., 69, 2038 (1965).
K. K. Sengupta, S. Sengupta and S. N. Basu, Carbohydr. Res., 71, 75 (1979); 80, 223 (1980); 80, 7 (1980); 79, 1 (1981).
A. Kumar and R. N. Mehrotra, J. Org. Chem., 40, 1248 (1975). (b) K. K. Sengupta and S. N. Basu, Carbohydr. Res., 72, 139 (1979).
C. R. Pottenger and D. C. Johnson, J. Polym. Sci., Part A-1, 8, 301 (1970); (b) R. N. Mehrotra and E. S. Emis, J. Org. Chem., 39, 1799 (1974).
H. S. Isbell and H. L. Frush, Carbohydr. Res., 28, 295 (1973).
T. Kistayya, M. S. Reddy and S. Kandlikar, Indian J. Chem. Sect. A., 25, 905 (1986).
M. Gupta, S. K. Saha and P. Banerjee, J. Chem. Soc., Perkin Trans. 1781 (1988).
H. S. Singh, B. Singh and A. K. Singh, Carbohydr. Res., 211, 235 (1991).
J. S. Mayell, Ind. Engng Chem., 7, 129 (1968).
W. P. Griffith, Q. Rev. Chem. Soc., 19, 254 (1965); (b) F. A. Cotton and G. Wilkinson, Advanced Inorganic Chemistry 2nd Edit., Wiley, Eastern Pvt. Ltd., New Delhi, 1966, p. 993 and 1007.
W. P. Griffith, The Chemistry of Rare Platinum Metals, Interscience, New York, 1967, pp. 42–125.
H. S. Singh in W. J. Mijs and C. R. H. I. De Jong (Eds.), Organic Synthesis by Oxidation with Metal Compounds, Plenum, New York, 1986, p. 633.
M. L. Wolform and W. L. Lewis, J. Am. Chem. Soc., 50, 837 (1928).
J. U. Nef., Annales., 403, 204 (1914); 356, 214 (1907).
F. Petuely and N. Meirner, Chem. Ber., 86, 1255 (1953).
P. Hirsch and R. Schlags Z. Physik. Chem. (Leipzig), A 141, 387 (1929).
K. B. Wiberg, Oxidation in Organic Chemistry, Academic Press, New York, 1965, Part ‘A’, p. 64.
C. H. Bamford and J. R. Collins, Proc. Roy. Soc. (London), A 204, 62 (1950); 204 85 (1950); 228, 100 (1955).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Singh, H.S., Gupta, A., Singh, A.K. et al. Kinetics and mechanism of the oxidation of reducing sugars by osmium tetroxide in alkaline medium. Transition Metal Chemistry 23, 277–281 (1998). https://doi.org/10.1023/A:1015756732447
Issue Date:
DOI: https://doi.org/10.1023/A:1015756732447