Abstract
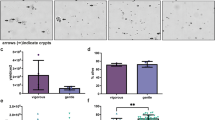
Some colonic luminal molecules resulting from bacterial metabolism of alimentary or endogenous compounds are believed to exert various effects on the colonic epithelial cell physiology. We isolated surface epithelial cells and intact colonic crypts in order to test bacterial metabolites in the pig model, which is often considered relevant for extrapolation to the physiopathology of the human gastrointestinal tract. Using colonocytes isolated with EDTA, we found that the initial cell viability, estimated by the membrane integrity and oxidative capacity measurement, fell rapidly despite several experimental attempts to preserve it such as the use of a medium designed to increase the adherence of epithelial cells and of a coated extracellular matrix, the presence in the culture medium of the oxidative substrate butyrate, and the use of an inhibitor of the caspases involved in cell apoptosis. In contrast, using dispase and collagenase as proteolytic agents, we were able to obtain pig colonic crypts that maintain an excellent membrane integrity after 4 h. Using this preparation, we were able to test the presumably cytotoxic luminal compounds hydrogen sulfide, ammonia, and deoxycholic acid on colonic crypt viability. Of these, only deoxycholic acid was found to significantly alter the cellular membrane integrity. It is concluded that pig colonic crypts can be useful for thein vitro appraisal of the cytotoxic properties of luminal compounds.
Similar content being viewed by others
References
Ahnen DJ. Etiology of large bowel cancer. In: Philipps S, Pemberton J, Shorter R, eds. The large intestine: physiology, pathology and disease. New York: Raven Press; 1991:501–20.
Ardawi MS, Newshoime EA. Fuel utilization in colonocytes of the rat. Biochem J. 1985; 231:713–9.
Bartram HP, Scheppach W, Schmid H, et al. Proliferation of human coionic mucosa as an intermediate biomarker of carcinogenesis: effects of butyrate, deoxycholate, calcium, ammonia, and pH. Cancer Res. 1993:53:3283–8.
Blachier F, Robert V, Selamnia M, Mayeur C, Duee PH. Sodium nitroprusside inhibits proliferation and putrescine synthesis in human colon carcinoma cells. FEBS Lett. 1996; 396:315–18.
Branka JE, Vallette G, Jarry A, et al. Early functional effects of Clostridium difficile toxin A on human colonocytes. Gastroenterology. 1997:112:1887–94.
Bull AW, Marnett LJ, Dawe EJ, Nigro ND. Stimulation of deoxythymidine incorporation in the colon of rats treated intrarectally with bile acids and fats. Carcinogenesis. 1983; 4:207–10.
Cheng H, Leblond CP. Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine. V. Unitarian theory of the origin of the four epithelial cell types. Am JAnat. 1974:141:537–61.
Chopra DP, Yeh KY. Long-term culture of epithelial cells from the normal rat colon. InVitro. 1981:17:441–9.
Darcy-Vrillon B, Morel MT, Cherbuy C, et al. Metabolic characteristics of pig colonocytes after adaptation to a high fiber diet. JNutr. 1993:123:234–43.
Evans GS, Flint N, Somers AS, Eyden B, Potten CS. The development of a method for the preparation of rat intest-inal epithelial cell primary cultures. J Cell Sci. 1992:101: 219–31.
Florin T, Neale G, Gibson GR, Christi SU, Cummings JH. Metabolism of dietary sulphate: absorption and excretion in humans. Gut. 1991:32:766–73.
Fry RJM, Staffeldt E. Effect of a diet containing sodium deoxycholate on the intestinal mucosa of the mouse. Nature. 1964:203:1396–8.
Goerg KJ, Specht W, Nell G, Rummel W, Schuiz L. Effect of deoxycholate on the perfused rat colon. Scanning and transmission electron microscopic study of the morphologi-cal alterations occurring during the secretagogue action of deoxycholate. Digestion. 1982:25:145–54.
Grossmann J, Maxson JM, Whitacre CM, et al. New isolation technique to study apoptosis in human intestinal epithelial cells. Am J Pathol. 1998a;153:53–62.
Grossmann J, Mohr S, Lapentina EG, Fiocchi C, Levine AD. Sequential and rapid activation of select caspases during apoptosis of normal intestinal epithelial cells. Am J Physiol. 1998b;274:GI 117–24.
Hass R, Busche R, Luciano L, Reale E, Engelhardt WV. Lack of butyrate is associated with induction of Bax and subse-quent apoptosis in the proximal colon of guinea pig. Gastroenterology. 1997:112:875–81.
Ichikawa H, Sakata T. Stimulation of epithelial cell prolifera-tion of isolated distal colon of rats by continuous coionic infusion of ammonia or short-chain fatty acids is nonadditive. JNutr. 1998:128:843–7.
Levitt MD, Fume J, Springfield J, Suarez F, DeMaster E. Detoxification of hydrogen sulfide and methanethiol in the cecal mucosa. J Clin Invest. 1999:104:1107–4.
Lewis ML, Moriarity DM, Campbell PS. Use of microgravity bioreactors for development of an in vitro rat salivary gland cell culture model. J Cell Biochem. 1993:51:265–73.
Lin HC, Visek WJ. Colon mucosal cell damage by ammonia in rats. JNutr. 1991a;121:887–93.
Lin HC, Visek WJ. Large intestinal pH and ammonia in rats: dietary fat and protein interactions. J Nutr. 1991b;121:832–43.
Lipkin M. Growth and development of gastrointestinal cells. Annu Rev Physiol. 1985:47:175–97.
Lowry OH, Rosebrough NJ, Farr AL, Randal RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951:193:265–75.
Luciano L, Hass R, Busche R, von Engelhardt W, Reale E. Withdrawal of butyrate from the coionic mucosa triggers “mass apoptosis” primarily in the GO/GI phase of the cell cycle. Cell Tissue Res. 1996:286:81–92.
Luft J. Improvements in epoxy resin embedding methods. J Biophys Biochem Cytol. 1961:9:409–14.
Macfarlane G, Cummings J. The coionic flora, fermentation and large bowel digestive function. In: Philipps S, Pemberton J, Shorter R, eds. The large intestine: physiology, pathology and disease. New York: Raven Press; 1991:51–92.
Macfarlane GT, Gibson GR, Cummings JH. Comparison of fermentation reactions in different regions of the human colon. JAppI Bacteriol. 1992:72:57–64.
Moore JW, Babidge W, Millard S, Roediger WE. Effect of sulphide on short chain acyl-CoA metabolism in rat colo-nocytes. Gut. 1997:41:77–81.
Mouille B, Morel E, Robert V, Guihot-Joubrel G, Blachier F. Metabolic capacity for L-citrulline synthesis from ammonia in rat isolated colonocytes. Biochim Biophys Acta. 1999; 1427:401–7.
Nicholls P. The effect of sulphide on cytochrome aa3. Isosteric and allosteric shifts of the reduced alpha-peak. Biochim Biophys Acta. 1975:396:24–35.
Pond W, Houpt K. The biology of the pig. lthaca: Cornell University Press; 1978:13–64.
Potten CS, Booth C, Pritchard DM. The intestinal epithelial stem cell: the mucosal governor. Int J Exp Pathol. 1997; 78:219–43.
Roediger WE. Role of anaerobic bacteria in the metabolic welfare of the coionic mucosa in man. Gut. 1980:21:793–8.
Roediger WE. Utilization of nutrients by isolated epithelial cells of the rat colon. Gastroenterology. 1982:83:424–9.
Roediger WE, Babidge W. Human colonocyte detoxification. Gut. 1997:41:731–4.
Roediger WE, Duncan A, Kapaniris O, Millard S. Reducing sulfur compounds of the colon impair colonocyte nutrition: implications for ulcerative colitis. Gastroenterology. 1993; 104:802–9.
Roediger WE, Moore J, Babidge W. Coionic sulfide in patho-genesis and treatment of ulcerative colitis. Dig Dis Sci. 1997:42:1571–9.
Sesink AL, Termont DS, Kleibeuker JH, Van der Meer R. Red meat and colon cancer: the cytotoxic and hyperproliferative effects of dietary heme. Cancer Res. 1999:59:5704–9.
Setchell KD, Lawson AM, Tanida N, Sjovall J. General methods for the analysis of metabolic profiles of bile acids and related compounds in feces. J Lipid Res. 1983:24:1085–100.
Sethi T, Rintoul RC, Moore SM, et al. Extracellular matrix proteins protect small cell lung cancer cells against apopto-sis: a mechanism for small cell lung cancer growth and drug resistance invivo. Nat Med. 1999:5:662–8.
Shao RG, Shimizu T, Pommier Y. 7-Hydroxystaurosporine (UCN-OI) induces apoptosis in human colon carcinoma and leukemia cells independently of p53. Exp Cell Res. 1997:234:388–97.
Strater J, Wedding U, Barth TF, Koretz K, Elsing C, Moller P. Rapid onset of apoptosis in vitro follows disruption of beta I-integrin/matrix interactions in human coionic crypt cellls. Gastroenterology. 1996:110:1776–84.
Suarez F, Fume J, Springfield J, Levitt M. Production and elimination of sulfur-containing gases in the rat colon. Am JPhysiol. 1998;274:G727–33.
Van der Meer R, Termont DS, DeVries HT. Differential effects of calcium ions and calcium phosphate on cytotoxicity of bile acids. Am J Physiol. 1991;260:G142–7.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Leschelle, X., Robert, V., Delpal, S. et al. Isolation of pig colonic crypts for cytotoxic assay of luminal compounds: Effects of hydrogen sulfide, ammonia, and deoxycholic acid. Cell Biol Toxicol 18, 193–203 (2002). https://doi.org/10.1023/A:1015515821390
Issue Date:
DOI: https://doi.org/10.1023/A:1015515821390