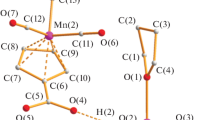
Abstract
Investigation on the nature of an initial mass loss observed for Mn(II)pyrrolidinedithiocarbamate complex is described. No evidences of decomposition are observed in infrared or mass spectra, elemental analysis and scanning electron microscopy of the residue at the end of the first thermal event, when compared with the same data for the original complex. Partial sublimation is assigned as the physical event after several experiments. The results are comparable with the findings for other complexes of the first transition series with pirrolidinedithiocarbamate.
Similar content being viewed by others
References
T. Kitson, Educ. Chem., (1985) 43.
R. M. Chaurasia, A. K. Sharma and S. K. Sharma, J. Ind. Chem. Soc., 58 (1981) 687.
S. T. Breviglieri, E. T. G. Cavalheiro and G. O. Chierice, Thermochim. Acta, 356 (2000) 79.
E. T. G. Cavalheiro, M. Ionashiro, G. Marino, S. T. Breviglieri and G. O. Chierice, Trans. Met. Chem., 25 (2000) 69.
A. R. Pray, Inorganic Synthesis, McGraw-Hill, New York 1957, p. 153.
D. D. Perrin and W. L. F. Armarego, Purification of Laboratory Chemicals, 3rd ed., Pergamon Press, New York 1988.
J. R. Dias, M. Zigon and S. Gomiscek, Org. Mass Spectrom., 16 (1981) 12.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Couto, A.B., Bernal, C., Chierice, G.O. et al. Initial Mass Loss in Anhydrous Manganese(II) Pyrrolidinedithiocarbamate. Decomposition or sublimation. Journal of Thermal Analysis and Calorimetry 67, 433–437 (2002). https://doi.org/10.1023/A:1013972410469
Issue Date:
DOI: https://doi.org/10.1023/A:1013972410469