Abstract
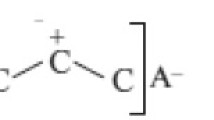
Products of liquid-phase oxidation of 2-methyl-2-butene and 2-methyl-2-pentene were studied by NMR and IR spectroscopy. Steric hindrances in liquid-phase oxidation of olefins with iso structure were shown to arise from branching of the alkyl group at the α-carbon atom.
Similar content being viewed by others
REFERENCES
Voronenkov, V.V. and Musabekov, Yu.S., Neftekhimiya, 1968, vol. 8, no. 5, pp. 713-717.
Nonhebel, D.C. and Walton, J.C., Free-Radical Chemistry, Cambridge: Cambridge Univ., 1974.
Kryukov, S.I., Yablonskii, O.P., Voronenkov, V.V., and Simanov, N.A., Zh. Org. Khim., 1971, vol. 7, no. 4, pp. 629-632.
Belyaev, V.A., Yablonskii, O.P., Voronenkov, V.V., and Pokrovskaya, Z.N., Neftekhimiya, 1973, vol. 13, no. 5, pp. 715-717.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Voronenkov, A.V., Simonov, N.A., Yablonskii, O.P. et al. Steric Factors in Liquid-Phase Oxidation of Olefins with iso Structure. Russian Journal of Organic Chemistry 37, 318–320 (2001). https://doi.org/10.1023/A:1012359819642
Issue Date:
DOI: https://doi.org/10.1023/A:1012359819642