Abstract
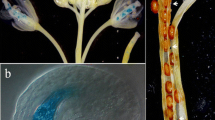
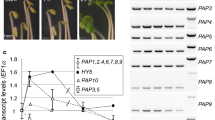
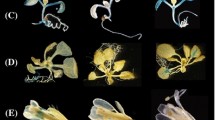
We describe an Arabidopsis thaliana gene, ptlpd2, which codes for a protein with high amino acid similarity to lipoamide dehydrogenases (LPDs) from diverse species. Ptlpd2 codes for a precursor protein possessing an N-terminal extension predicted to be a plastid-targeting signal. Expression of the ptlpd2 cDNA in Escherichia coli showed the encoded protein possessed the predicted LPD activity. PTLPD2 protein, synthesized in vitro, was efficiently imported into isolated chloroplasts of Pisum sativum and shown to be located in the stroma. In addition, fusion proteins containing the predicted transit peptide of PTLPD2 or the entire protein fused at the N-terminus with the green fluorescent protein (GFP), showed accumulation in vivo in chloroplasts but not in mitochondria of A. thaliana. Expression of ptlpd2 was investigated by introducing ptlpd2 promoter-β-glucuronidase (GUS) gene fusions into Nicotiana tabacum. GUS expression was observed in seeds, flowers, root tips and young leaves. GUS activity was highest in mature seeds, decreased on germination and increased again in young leaves. Expression was also found to be temporally regulated in pollen grains where it was highest in mature grains at dehiscence. Database searches on ptlpd2 sequences identified a second A. thaliana gene encoding a putative plastidial LPD and two genes encoding proteins with high similarity to the mitochondrial LPD of P. sativum.
Similar content being viewed by others
References
Altschul, S.F., Madden, T.L., Schaffer, A.A., Zhang, J., Zhang, Z., Miller, W., and Lipman, D.J. 1997. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucl. Acids Res. 25: 3389–3402.
Bao, X., Focke, M., Pollard, M. and Ohlrogge, J. 2000. Understanding in vivo carbon precursor supply for fatty acid biosynthesis in leaf tissue. Plant J. 22: 39–50.
Benen, J.A.E., van Berkel, W.J.H., Veeger, C. and de Kok, A. 1992. Lipoamide dehydrogenase from Azotobacter vinelandii:the role of the C-terminus in catalysis and dimer stabilization. Eur. J. Biochem. 207: 495–505.
Bevan, M. 1984. Binary Agrobacterium vectors for plant transformation. Nucl. Acids Res. 12: 8711–8721.
Bevan, M., Bancroft, I., Bent, E. et al. 1998. Analysis of 1.9 Mb of contiguous sequence from chromosome 4 of Arabidopsis thaliana. Nature 391: 485–488.
Bourguignon, J., Macheral, D., Neuberger, M. and Douce, R. 1992. Isolation, characterization and sequence analysis of a cDNA clone encoding L-protein, the dihydrolipoamide dehydrogenase component of the glycine cleavage system from pea-leaf mitochondria. Eur. J. Biochem. 204: 865–873.
Bourguignon, J., Merand, V., Rawsthorne, S., Forest, E. and Douce, R. 1996. Glycine decarboxylase and pyruvate dehydrogenase complexes share the same dihydrolipoamide dehydrogenase in pea leaf mitochondria: evidence from mass spectrometry and primary-structure analysis. Biochem. J. 313: 229–234.
Bowman, S.B., Zaman, Z. Collinson, L.P., Brown, A.J. and Dawes, I.W. 1992. Positive regulation of the LPD1 gene of Saccharomyces cerevisiae by the HAP2/HAP3/HAP4 activation system. Mol. Gen. Genet. 231: 296–303.
Bradford, M.M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–255.
Busk, P.K. and Pagès, M. 1998. Regulation of abscisic acid-induced transcription. Plant Mol. Biol. 37: 425–435.
Camp, P.J. and Randall, D.D. 1985. Purification and characterization of the pea chloroplast pyruvate dehydrogenase complex. Plant Physiol. 77: 571–577.
Conner, M., Krell, T. and Lindsay, J.G. 1996. Identification and purification of a distinct dihydrolipoamide dehydrogenase from pea chloroplasts. Planta 200: 195–202.
Dastoor, F.P., Forrest, M.E. and Beatty, J.T. 1997. Cloning, se-quencing and oxygen regulation of the Rhodobacter capsulatus alpha-ketoglutarate dehydrogenase operon. J. Bact. 179: 4559–4566.
Davis, S.J. and Vierstra, R.D. 1998. Soluble, highly fluorescent variants of green fluorescent protein (GFP) for use in higher plants. Plant Mol. Biol. 36: 521–528.
Denyer, K. and Smith, A.M. 1988. The capacity of plastids from developing pea cotyledons to synthesize acetyl-CoA. Planta 173: 172–182.
Emanuelsson, O., Nielsen, H. and von Heijn, G. 1999. ChloroP, a neural network-based method for predicting chloroplast transit peptides and their cleavage sites. Protein Sci. 8: 978–984.
Engels, A. and Pistorius, E.K. 1997. Characterization of a gene encoding dihydrolipoamide dehydrogenase of the cyanobacterium Synechocystis sp. strain PCC 6803. Microbiology 143: 3543–3553.
Faure, M., Bourguignon, J., Neuburger, M., Macherel, D., Sieker, L., Ober, R., Kahn, R., Cohen-Addad, C. and Douce, R. 2000. Interaction between the lipoamide-containing H-protein and the lipoamide dehydrogenase (L-protein) of the glycine de-carboxylase multienzyme system. 2. Crystal structures of H-and L-proteins. Eur. J. Biochem. 267: 2890–2898.
Foster, R., Izawa, T. and Chua N.H. 1994. Plant b-ZIP proteins gather at AGCT elements. FASEB J. 8: 192–200.
Gavel, Y. and von Heijne, G. 1990. A conserved cleavage-site motif in chloroplast transit peptides. FEBS Lett. 261: 455–458.
Gray, M.W. 1999. Evolution of organellar genomes. Curr. Opin. Genet. Dev.9: 678–687.
Harwood, J.L. 1996. Recent advances in biosynthesis of fatty acids. Biochim. Biophys. Acta 1301: 7–56.
Hibberd, J.M, Linley, P.J., Khan, M.S. and Gray, J.C. 1998. Transient expression of green fluorescent protein in various plastid types following microprojectile bombardment. Plant J. 16: 627–632.
Higo, K., Ugawa, T., Iwamoto, M. and Higo, H. 1998. PLACE: a database of plant cis-acting regulatory DNA elements. Nucl. Acids Res. 26: 358–359.
Horsch, R.B., Fry, J.E., Hoffman, N.L., Eicholtz, D., Rogers, S.G. and Fraley, R.T. 1985. A simple and general method for transferring genes into plants. Science227: 1229–1231.
Jefferson, R.A., Kavanagh, T.A. and Bevan, M.W. 1987. GUS fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J. 6: 3901–3907.
Johnston, M.L., Luethy, M.H., Miernyk, J.A. and Randall, D.D. 1997. Cloning and molecular analyses of the Arabidopsis thaliana plastid pyruvate dehydrogenase subunits. Biochim. Biophys. Acta 1321: 200–206.
Johnston, M.L., Miernyk J.A. and Randall, D.D. 2000. Import, processing, and assembly of the α-and β-subunits of chloroplast pyruvate dehydrogenase. Planta 211: 72–76.
Kang, F. and Rawsthorne, S. 1994. Starch and fatty acid synthesis in plastids from developing embryos of oilseed rape (Brassica napus L.). Plant J. 6: 795–805.
Ke, J., Behal, R.H., Back, S.L., Nikolau, B.J., Wurtele, E.S. and Oliver, D.J. 2000. The role of pyruvate dehydrogenase and acetyl-coenzyme A synthase in fatty acid synthesis in developing Arabidopsis seeds. Plant Physiol. 123: 497–508.
Kimura, M. 1980. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequence. J. Mol. Evol. 16: 111–120.
Lutziger, I. and Oliver, D.J. 2000. Molecular evidence of a unique lipoamide dehydrogenase in plastids: analysis of plastidic lipoamide dehydrogenase from Arabidopsis thaliana. FEBS Lett. 484: 12–16.
Mattevi, A., Schierbeek, A.J. and Hol, W.G. 1991. The refined crystal structure of Azotobacter vinelandii lipoamide dehydrogenase at 2.2 Å resolution. A comparison with the structure of glutathione reductase. J. Mol. Biol. 220: 974–995.
Mattevi, A., Obmolova, G., Kalk, K.H., van Berkel, W.J. and Hol, W.G. 1993. Three-dimensional structure of lipoamide dehydrogenase from Pseudomonas fluorescens at 2.8 Å resolution. Analysis of redox and thermostability properties. J. Mol. Biol. 230: 1200–1215.
Mooney, B.P., Miernyk, J.A. and Randall, D.D. 1999. Cloning and characterization of the dihydrolipoamide S-acetyltransferase subunit of the plastid pyruvate dehydrogenase complex (E2) from Arabidopsis. Plant Physiol. 120: 443–452.
Mould, R.M. and Gray, J.C.1998a. Preparation of chloroplasts for protein synthesis and protein import. In: J.E. Celis (Ed.) Cell Biology: A Laboratory Handbook, vol. 2, Academic Press, New York, pp. 81–86.
Mould, R.M. and Gray, J.C. 1998b. Import of nuclear-encoded proteins by isolated chloroplasts and thylakoids. In: J.E. Celis (Ed.) Cell Biology: A Laboratory Handbook, vol. 2, Academic Press, New York, pp. 286–292.
Nakai, K. and Kanehisa, M. 1992. A knowledge base for predicting protein localization sites in eukaryotic cells. Genomics. 14: 897–911.
Patel, M.S. and Roche, T.E. 1990. Molecular biology and biochemistry of pyruvate dehydrogenase complexes. FASEB J. 4: 3224–3233.
Stevens R.G., Creissen, G.P. and Mullineaux, P.M. 1997. Cloning and characterization of a cytosolic glutathione reductase cDNA from pea (Pisum sativum L.) and its expression in response to stress. Plant Mol. Biol. 35: 641–654.
Tatusova, T.A. and Madden, T.L. 1999. BLAST 2 sequences: a new tool for comparing protein and nucleotide sequences. FEMS Microbiol. Lett. 174: 247–250.
Taylor, A.E., Cogdell, R.J. and Lindsay, J.G. 1992. Immunological comparison of the pyruvate dehydrogenase complexes from pea mitochondria and chloroplasts. Planta 188: 225–231.
Toyoda, T., Suzuki, K., Sekiguchi, T., Reed, L.J. and Takaneka, A. 1998. Crystal structure of eukaryotic E3, lipoamide dehydrogenase from yeast. J. Biochem. 123: 668–674.
Twell, D., Yamaguchi, J., Wing, R.A., Ushiba, J. and McCormick, S. 1991. Promoter analysis of genes that are coordinately expressed during pollen development reveals pollen-specific enhancer sequences and shared regulatory elements. Genes Dev. 5: 496–507.
Zaman, Z., Bowman, S.B., Kornfeld, G.D., Brown, A.J. and Dawes, I.W. 1999. Transcription factor GCN4 for control of amino acid biosynthesis also regulates the expression of the gene for lipoamide dehydrogenase. Biochem. J. 340: 855–862.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Drea, S.C., Mould, R.M., Hibberd, J.M. et al. Tissue-specific and developmental-specific expression of an Arabidopsis thaliana gene encoding the lipoamide dehydrogenase component of the plastid pyruvate dehydrogenase complex. Plant Mol Biol 46, 705–715 (2001). https://doi.org/10.1023/A:1011612921144
Issue Date:
DOI: https://doi.org/10.1023/A:1011612921144