Abstract
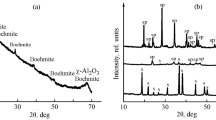
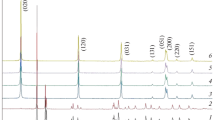
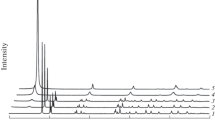
The formation process of bayerite, from an aqueous solution of sodium aluminate through enforced decomposition of aluminate ions by introducing CO2 gas and aging with mechanical stirring, was investigated by pH measurements of the mother solution during preparation reaction and characterization of precipitates obtained at various stages of preparation. An amorphous precipitate, produced initially by the reaction of introduced CO2, transformed to bayerite via pseudoboehmite during aging. It was found that the crystalline particle size and morphology of the crystallized bayerite change depending systematically on the preparation conditions. The reaction pathway of the thermal decomposition of the synthesized bayerite was investigated by using thermoanalytical techniques.
Similar content being viewed by others
References
Hector Juarez M., J. Merced Martinez R., J. Manuel Ruvalcaba L., Oriana A. Vargas P. and Juan Serrato R., Am. Ceram. Soc. Bull., 76 (1997) 55.
H. C. Stumpf, Ind. Eng. Chem., 42 (1950) 1398.
V. H. Ginsberg, W. Huttig and H. Stiehl, Z. anorg. allg. Chem., 309 (1961) 16.
M. Pyzalsky, M. Wojcik and R. Urbansky, Light Met., (1992) 131.
A. van Straten, B. T. W. Holtkamp and P. L. de Bruyn, J. Colloid Interface Sci., 98 (1984) 348.
T. Sato, J. Appl. Chem., 12 (1962) 553.
T. Tsuchida and N. Ichikawa, React. Solids, 7 (1989) 207.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Koga, N., Fukagawa, T. & Tanaka, H. Preparation and Thermal Decomposition of Synthetic Bayerite. Journal of Thermal Analysis and Calorimetry 64, 965–972 (2001). https://doi.org/10.1023/A:1011531215596
Issue Date:
DOI: https://doi.org/10.1023/A:1011531215596