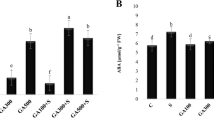
Abstract
Literature data point to a possible link between gibberellic acid (GA3) and glutathione metabolism in plant tissue, as both are connected to dormancy breakage. In order to study the influence of GA3 on glutathione metabolism, we treated an anthocyanin accumulating cell culture of periwinkle (Catharanthus roseus) and a shoot differentiated culture of pea (Pisum sativum) with GA3. Glutathione reductase (GR; E.C. 1.6.4.2) activity increased to 135% and 190% of the control in C. roseus and P. sativum, respectively. The level of oxidized glutathione (GSSG) decreased to 60% of the control in the C. roseus culture while no change in GSSG was observed in the P. sativum culture. No changes in the tissue concentration of total glutathione was observed in the cultures after GA3 treatment. Concomitant to the changes in GSSG and GR, an increase in anthocyanin accumulation was observed in the C. roseus culture in association with a strong increase in phenylalanine ammonia-lyase (PAL; E.C. 4.3.1.5) activity in response to GA3. These data strongly suggest a link between GA3 and glutathione metabolism.
Similar content being viewed by others
References
Berglund T & Ohlsson AB (1995) Defensive and secondary metabolism in plant tissue cultures, with special reference to nicotinamide, glutathione and oxidative stress. Plant Cell Tiss. Org. Cult. 43: 137–145
Berglund T, Ohlsson AB & Rydström J (1993) Nicotinamide increases glutathione and anthocyanin in tissue culture of Catharanthus roseus. J Plant Physiol 141: 596–600
Bielawski W & Joy KW (1986) Properties of glutathione reductase from chloroplasts and roots of pea. Phytochemistry 25: 2261–2265
Bolwell GP (1985) Use of tissue cultures for studies on vascular differentiation. In: Dixon RA (ed) Plant Cell Culture: a Practical Approach (pp 107–126). IRL Press, Oxford
Edwards EA, Rawsthorne S & Mullineaux PM (1990) Subcellular distribution of multiple forms of glutathhione reductase in leaves of pea (Pisum sativum) L.). Planta 180: 278–284
Foyer CH, Lopez–Delgado H, Dat JF & Scott SM (1997) Hydrogen peroxide–and glutathione–associated mechanisms of acclimatory stress tolerance and signalling. Physiol Plant. 100: 241–254
Huttly AK & Phillips AL (1995) Gibberellin–regulated plant genes. Physiol. Plant. 95: 310–317
Kranner I & Grill D (1996) Significance of thiol–disulfide exchange in resting stages of plant development. Bot. Acta 109: 8–14
Kurepa J, Hérouart D, Van Montagu M & Inzé D (1997) Differential expression of CuZn–and Fe–superoxide dismutase genes of tobacco during development, oxidative stress, and hormonal treatments. Plant Cell Physiol. 38: 463–470
Marrs KA, Alfenito MR, Lloyd AM & Walbot V (1995) A glutathione S–transferase involved in vacuolar transfer encoded by the maize gene Bronze–2. Nature 375: 397–400
Ohlsson AB & Björk L (1988) Effects of gibberellic acid on cardenolide accumulation by Digitalis lanata tissue cultures grown in light and darkness. J. Plant Physiol. 133: 535–538
Petersen GL (1977) A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal. Biochem. 83: 346–356
Smith IK, Vierheller TL & Thorne CA (1988) Assay of glutathione reductase in crude tissue homogenates using 5,5'–dithiobis(2–nitrobenzoic acid). Anal. Biochem. 175: 408–413
Wang SY, Jiao HJ & Faust M (1991) Changes in ascorbate, glutathione, and related enzyme activities during thidiazuron–induced bud break of apple. Physiol. Plant 82: 231–236
Weiss D, van Blokland R, Kooter JM, Mol JNM & van Tunen AJ (1992) Gibberellic acid regulates chalcone synthase gene transcription in the corolla of Petunia hybrida. Plant Physiol. 98: 191–197
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ohlsson, A.B., Berglund, T. Gibberellic acid-induced changes in glutathione metabolism and anthocyanin content in plant tissue. Plant Cell, Tissue and Organ Culture 64, 77–80 (2001). https://doi.org/10.1023/A:1010687811222
Issue Date:
DOI: https://doi.org/10.1023/A:1010687811222